

133 Molesworth
Street
PO Box 5013
Wellington 6140
New Zealand
T+64 4 496 2000
21 June 2021
Andrew Lewis
By email:
[FYI request #15147 email]
Ref:
H202107070
Dear Andrew Lewis
Response to your request for official information Thank you for your follow up request under the Official Information Act 1982 (the Act) to the
Ministry of Health (the Ministry) on 27 May 2021 for:
“…please supply the Comirnaty vaccine package insert information.”
The instructions for the preparation and administration of the Pfizer/BioNTech vaccine as at
11 June 2021 is enclosed.
I trust this information fulfils your request. Under section 28(3) of the Act you have the right
to ask the Ombudsman to review any decisions made under this request. The Ombudsman
may be contacted by email at: [email address] or by calling 0800 802 602.
Please note that this response, with your personal details removed, may be published on the
Ministry website at: www.health.govt.nz/about-ministry/information-releases/responses-
official-information-act-requests.
Yours sincerely
Joanne Gibbs
National Director
COVID-19 Immunisation Vaccine Programme




Pfizer New Zealand Limited Level 1,
Suite 1.4, Building B
8 Nugent Street, Grafton, Auckland 1023
PO Box 3998, Shortland Street, Auckland, New Zealand 1140
Tel: 09 354 3065 Fax: 09 374 7630
Pfizer New Zealand Limited
24 March 2021
Dear Healthcare Professional,
Supply of COMIRNATY COVID-19 Vaccine in New Zealand
Pfizer New Zealand Limited has commenced supply of COMIRNATY COVID-19 VACCINE 0.5 mg/mL
concentrated suspension for injection. COMIRNATY is indicated for active immunisation to
prevent coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2, in individuals 16 years of
age and older. The use of this vaccine should be in accordance with official recommendations.
As the national COVID-19 immunisation plan is being implemented as quickly as possible, it is
appropriate that important practical information regarding this vaccine is understood.
Identification and label ing
Pfizer’s COVID-19 vaccine is supplied as a concentrated suspension in a 2 mL, multi-dose glass
vial with a purple, flip-off cap. The vaccine wil be supplied in trays containing 195 vials, or other
packaging configurations depending upon the agreement with the New Zealand government.
Due to the nature of the pandemic, production and supply of the vaccine is being managed in a
just-in-time model, which means that it is possible to receive the vaccine packaged in a number
of different artworks. Given the name of the vaccine and the name of the active ingredient have
changed over time, it is possible that the fol owing names may appear on labels, inserts and
other associated packaging components:
Product name:
Pfizer-BioNTech COVID-19 Vaccine
under the Official Information Act 1982
COVID-19 Vaccine
COVID-19 mRNA Vaccine
COMIRNATY
Active ingredient (generic) name:
BNT162b2 [mRNA]
Irrespective of the pack livery, al are equivalent, in that they refer to the same product with
Released
the same formulation.
Example product label ing is attached to this letter. Please be aware of the fol owing:
1. ‘Pfizer-BioNTech COVID-19 Vaccine’ (see Attachment 1):
The labels wil also state that they are for use under Emergency Use Authorization. This
statement has been included to meet the requirements of the US Food and Drug
Administration (FDA) but is not relevant or applicable to the vaccine’s use in New Zealand.
The labels wil state that the vaccine MUST BE DILUTED BEFORE USE with sterile 0.9%
Sodium Chloride Injection, USP. However, any pharmacopeial grade of sterile 0.9% sodium
chloride can be used for dilution of this vaccine.
The use of the name ‘BNT 162b2 (SARS-COV-2-mRNA vaccine) 5-dose vial’ in the fact sheet is
not applicable to NZ. Vaccine supplied in NZ wil be referred to differently, as explained
1982
above).
The vials instruct to record the Date and Time of dilution, this is aligned with the instructions
Act
in the Medsafe-approved Data Sheet.
2. Product supplied with the tradename ‘COMIRNATY’ (see Attachment 2):
Instructions on the vials require the Date and Time that the vial contents should be
discarded to be recorded on the vial. This is different to the instructions in the Medsafe
approved Data Sheet, which instruct to record the Date and Time of dilution.
Number of doses per vial
This is a multidose vial and must be diluted with 1.8 mL 0.9% saline solution before use.
Instructions for dilution are contained in the Medsafe approved Data Sheet available at
Information
www.medsafe.govt.nz/profs/Datasheet/c/comirnatyinj.pdf. One vial (0.45 mL) contains 6 doses
of 0.3 mL after dilution. 1 dose (0.3 mL) contains 30 micrograms of BNT162b2 [mRNA]
(embedded in lipid nanoparticles).
Whilst some stock is label ed as containing 5 doses when diluted, 6 doses may be withdrawn
from each vial, if the appropriate combination of low dead-volume needles and/or syringes is
Official
used. This information is reflected in the Medsafe-approved Data Sheet. If standard syringes and
needles are used, there may not be sufficient volume to extract a sixth dose. If the amount of
vaccine remaining in the vial cannot provide a ful 0.3 mL dose, discard the vial and any excess
the
volume. DO NOT pool excess vaccine from multiple vials.
Each box of the vaccine may contain either the US fact sheet or the EU fact sheet as a package
insert. For the purpose of use in New Zealand, please refer to the Medsafe-approved Data Sheet
for COMIRNATY that is available on the Medsafe website.
under
Storage requirements for the frozen, thawed and diluted vaccine
Adherence to the storage and handling guidance relating to the vaccine is critical to ensuring
its quality and efficacy. As al of the storage guidance may not be present on the labels
supplied, please ensure that the fol owing guidance is fol owed when storing the vaccine. This
guidance is also provided in the Medsafe-approved Data Sheet and the resources provided by
Pfizer.
Released
Frozen vaccine
The vaccine is shipped frozen to New Zealand and should be stored at -90°C to -60°C. Unopened
vials may also be stored and transported at -25°C to -15°C for a total of 2 weeks on one occasion
only and can then be returned to -90ºC to -60°C. The vaccine should be kept in the original
package in order to protect it from light. During storage, take care to minimise exposure of the
vaccine to room light, and avoid exposure to direct sunlight and ultraviolet light. Refer to sections
6.3 and 6.4 of the Data Sheet for additional information on handling the frozen vial trays.



Thawed vaccine
The vaccine must be thawed prior to dilution. Frozen vials should be transferred to an
environment of 2°C to 8°C to thaw; a 195 vial pack may take 3 hours to thaw. Alternatively,
frozen vials may also be thawed for 30 minutes at temperatures up to 30°C for immediate use.
Once removed from the freezer, the unopened vaccine can be stored for up to 5 days at 2°C to
1982
8°C, and up to 2 hours at temperatures up to 30 °C, prior to use. Thawed vials can be handled in
room light conditions. Once thawed, COMIRNATY should not be re-frozen.
Act
Depending upon your location, you could receive the vials pre-thawed at 2°C to 8°C. Vials that
have been pre-thawed at a site approved by the New Zealand Ministry of Health wil be
repackaged in new cartons containing 5 or 15 vials per carton, and wil be label ed with the
thawed expiry date (5 days from the date of thawing). If you receive the vaccine pre-thawed,
please note the expiry date that is marked on the carton label, as this represents the date that
the vaccine must be diluted and administered by. Refer to section 6.4 of the Data Sheet for
information regarding the storage of thawed vaccine and diluted vaccine.
Diluted vaccine
Information
For diluted medicinal product, chemical and physical in-use stability has been demonstrated for 6
hours at 2°C to 30°C after dilution in sodium chloride 9 mg/mL (0.9%) solution for injection. From
a microbiological point of view, the product should be used immediately. If not used
immediately, microbiological risks and package integrity are the responsibility of the user.
For additional information regarding Pfizer’s COVID-19 vaccine, refer to the COMIRNATY Data
Official
Sheet on the Medsafe website www.medsafe.govt.nz/profs/Datasheet/c/comirnatyinj.pdf, or
contact Pfizer by phone (0800 736 363) or e-mail [email address]
the
Scott Wil iams
Vaccines Medical Director New Zealand, Australia and Korea
under
Released

 Instructions for multi-dose vial
Pfizer/BioNTech vaccine:
preparation and administration
Instructions for multi-dose vial
Pfizer/BioNTech vaccine:
preparation and administration 1982
STORAGE
Act
Pfizer vaccine vials will thaw during distribution to clinics. DO NOT REFREEZE.
Store undiluted vials at +2°C to +8°C for up to 31 days. This includes distribution time.
For dilution, vials can be stored at room temperature (but less than +30°C) for an additional two hours at most.
Pfizer vaccine comes as a concentrate and MUST be diluted.
After dilution, store vials between +2°C and +30°C and use within six hours.
You must monitor room temperature.
Avoid exposure of vaccine to direct sunlight, or UV light at any time.
PRIOR TO DILUTION
1. Before dilution, the vaccine should be brought to room temperature i.e. It should not
feel cold to touch.
Information
2. Before dilution, invert the vaccine vial gently
10 times.
Gently x 10
3.
DO NOT SHAKE.
4.
Inspect liquid in vial prior to dilution. The thawed suspension may contain white to
off‑white opaque amorphous particles.
DILUTING THE VACCINE
Official
1. Remove metal cap guard on top of vaccine vial using non‑touch technique.
2. Cleanse vaccine vial stopper with a single‑use antiseptic swab and leave to dry.
the
3. Use
supplied diluent (0.9% sodium chloride for injection).
4. Open diluent just before use. Diluent that is opened and not used immediately
must be discarded.
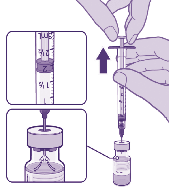
5. Using aseptic technique, withdraw
1.8 mL of diluent into a syringe (using a
25‑gauge orange standard needle).
1.8mL of 0.9%
sodium chloride for injection
under
Slowly add 1.8 mL of diluent into the vaccine vial.
EQUALIZE
• Before withdrawing any fluid, equalize vial pressure by
withdrawing 1.8 mL of air
into the now empty diluent syringe.
• Remove the syringe and needle attached to the vaccine vial and discard.
• After dilution, the vial contains 2.25 mL from which 6 doses of 0.3 mL
can be extracted
Released
Pull back plunger to 1.8mL
to remove air from vial
AFTER DILUTION
1.
Gently invert vial containing diluted vaccine
10 times to mix.
2.
DO NOT SHAKE.
.0
3. Inspect vaccine in vial. It should be an off‑white suspension with no particulates visible.
4. Do not use if vaccine is discoloured or contains particulate matter.
4/05/2021 v1
C 2
IMA
INSTRUCTIONS FOR MULTI-DOSE VIAL PFIZER/BIONTECH VACCINE: PREPARATION AND ADMINISTRATION
1

 LABELLING VACCINE
LABELLING VACCINE
• Record
date and
time of dilution on the vaccine vial label.
• Vaccine must be used within 6 hours and can be stored at room temperature.
If returned to the fridge, allow to return to room temperature, i.e. not cold
to touch, before administration.
• The 6‑hour allowance for use is in addition to the 31 days of distribution and storage
1982
at +2°C to +8°C.
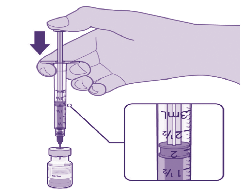
PREPARATION OF INDIVIDUAL 0.3mL DOSES OF PFIZER/BIONTECH
Act
Each dose is drawn up using low dead space (LDS) needles that they will be
0.3 mL
administered with.
dilute vaccine
We recommend drawing up all doses immediately. Syringe and needles are only
removed from their packaging just before use.
1. Cleanse the vial stopper with a single‑use antiseptic swab.
2. Using aseptic technique, withdraw 0.3 mL of vaccine using a 1 mL syringe with
an LDS Long Orange Needle 25g x 25mm (1”) attached.
3. This vaccine is now ready for patient administration,
do not swap the needle.
This needle will be used to administer the vaccine to the patient.
Information
4. Unless you plan to administer immediately, carefully replace the needle guard
and place syringe onto a ridged tray for storage.
5. Draw up the next dose of vaccine in the same manner. It is not necessary to swab the top
of the vial between drawing up each dose of vaccine if drawn up one after the other.
• Pierce the stopper in a different place each time. Imagining clock face positions
Official
on the stopper works well‑ insert the needles at 2 o’clock, 4 o’clock, 6 o’clock etc
around the stopper.
• Draw the dose up slowly and carefully to reduce risk of air bubbles.
the
• Each dose must contain 0.3 mL of vaccine
If vial is left for any time, i.e. while administering the vaccine, then the top will need to
be cleansed and time allowed for the alcohol to dry, before the next dose is withdrawn.
The expected number of doses per diluted vaccine vial is usually six. If the amount of
vaccine remaining in the vial cannot provide another full 0.3 mL dose, discard vial and
under
remaining diluted vaccine.
Do not pool excess vaccine from multiple vials.
Discard any unused vaccine six hours after dilution.
If low dead space needles are not available, you can still administer vaccines
as above but using standard needles. For larger patients you may need to use 23‑25G x 1.5” 38mm length. Draw up
and administer as above.
Released
ADMINISTRATION
Visually inspect each dose in each syringe
prior to administration:
• check volume is
0.3 mL
• confirm there is
no discoloration or particulate matter.
Do not administer if vaccine is discoloured or contains particulate matter.
.0
Administer vaccine intramuscularly into the deltoid.
4/05/2021 v1
C 2
IMA
INSTRUCTIONS FOR MULTI-DOSE VIAL PFIZER/BIONTECH VACCINE: PREPARATION AND ADMINISTRATION
2
Document Outline













