 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
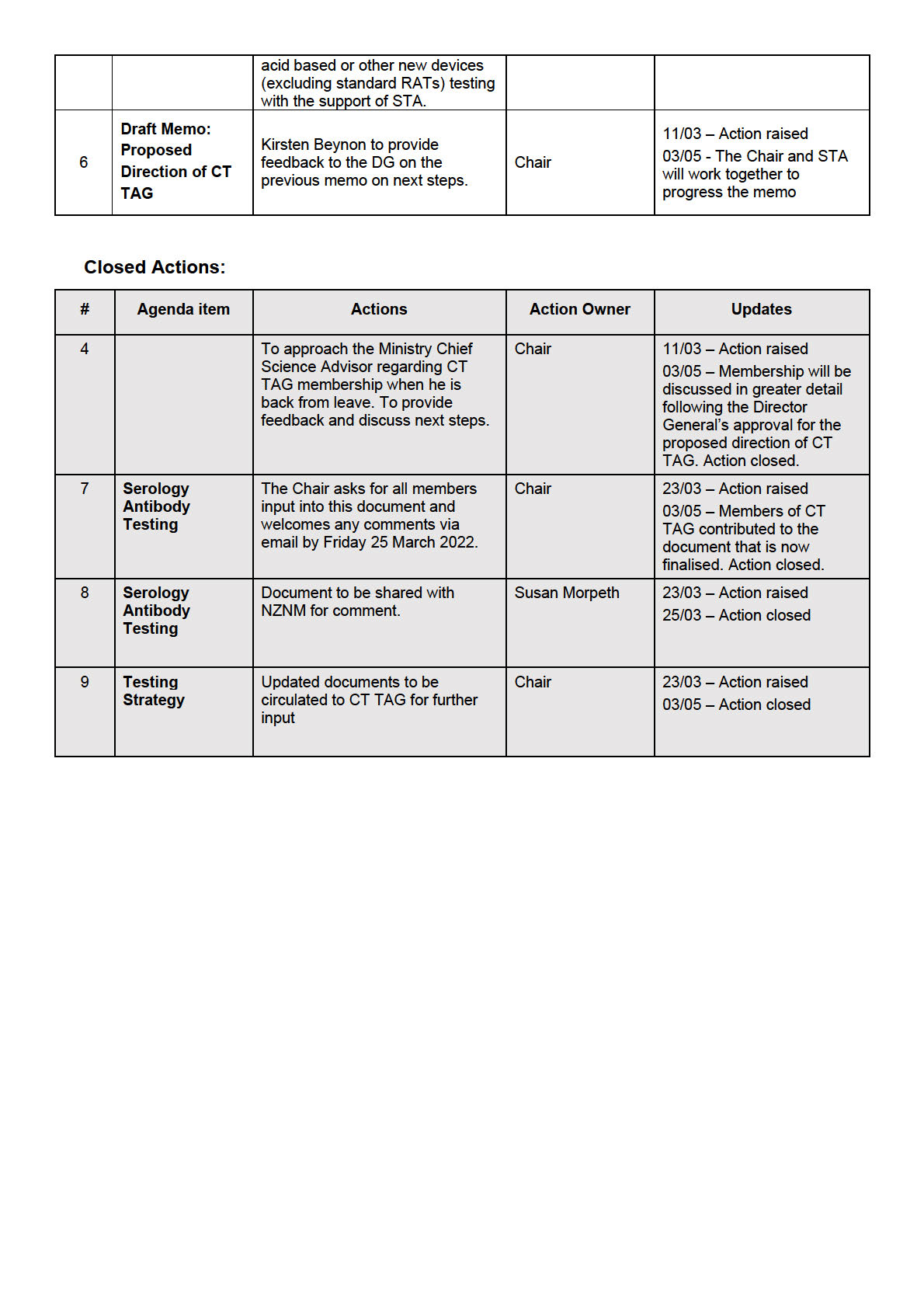
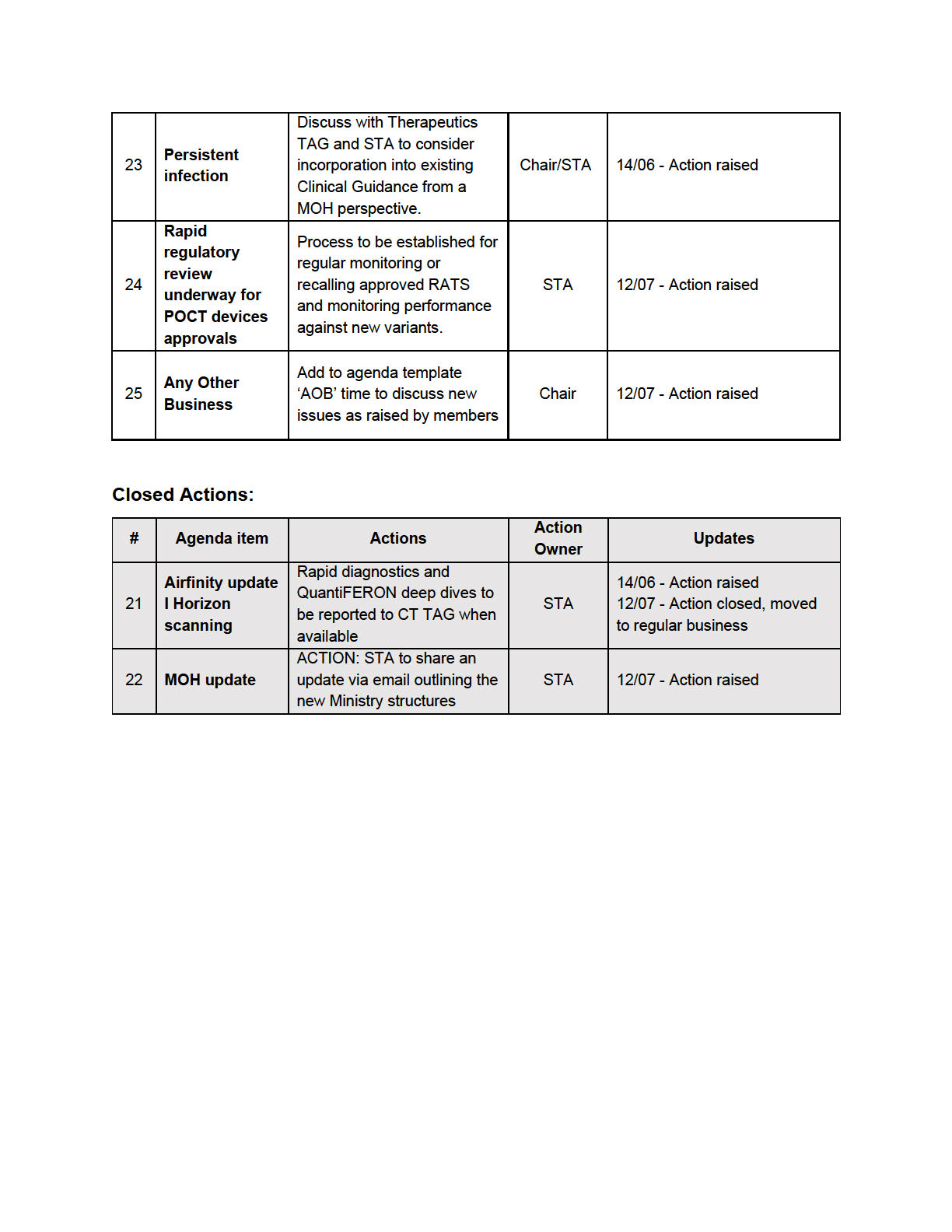
• The Chair asked for the documentation informing the Plan to be shared with the group through
email to give further clarity of the test modalities and rationale behind it.
• It was noted that the CT TAG members could give valuable input on the ongoing use of RATs in a
New Zealand context to support decision making, recognising that tests are imperfect, and
implementation wil look different for different scenarios.
• CT TAG members advice on the Implementation Plan would be welcomed. This wil be
communicated through email to the members of the Group and wil focus on the more granular
details needed for health care providers.
• The members were encouraged to bring questions and feedback to the following CT TAG
meeting.
ACTION: CT TAG to support the Testing team to form an Implementation Plan.
4.0
SARS-CoV-2 Antibody and Immunity Testing Memo
• The Chair thanked the TAG for their input into this document.
1982
• This paper has supported conversations in the space, and is now being shaped into a Health
Report to be utilised more widely. The members of the group wil receive a copy of this once
finalised.
ACT
5.0
Draft Memo: Proposed Direction of CT TAG
• The Ministry apologised for the memo taking longer than anticipated and highlighted the members
feedback is being collated.
ACTION UPDATE: The Chair and STA wil work together to progress the memo to DG.
6.0
Testing Plan
Discussed in agenda item 3.0
INFORMATION
7.0
Surveil ance Plan
• It was noted there was little information surrounding the timeline for implementation. In general,
the Plan was seen to have been oversimplified and overly broad. It was agreed that further clarity
on implementation was needed.
OFFICIAL
• Whole genome sequencing on al imported cases could be difficult to execute. A member noted
the rationale for whole genome sequencing was not included in the document and there is
uncertainty of how new variants w
THE ould be detected.
• It was suggested ESR should link into routine diagnostics. CT TAG would like to clarify with ESR
what they are doing with regards to new variants and seek input into the surveil ance design.
ACTION: Ministry to invite ESR to a CT TAG meeting to give input into the surveil ance design.
UNDER
8.0
COVID-19 Therapeutics TAG: Use of Serology
• The ability to perform serologic testing is currently under discussion for diagnosing those with
persistent COVID infection, and this would aid discussions on eligibility for convalescent plasma or
monoclonal antibody therapies. It was noted that it would be useful for both prevention and
treatment of persistent COVID infection in the immunosuppressed.
• A question was raised on how to interpret the result for those that return a low serological
response.
RELEASED
• The Ministry noted the importance of this to be included in Hospital Guidelines.
• It was agreed that there is the need to consider broadening who is eligible to donate blood. This
will aim to increase the supply for convalescent plasma treatments.
• It was suggested that the Ministry contact the New Zealand Blood service and request broadening
eligible donations to include those who lived in the United Kingdom between 1980 and 1996. It
was noted they could also be contacted about conducting antibody testing for immunoglobulin
therapy in IVIg products, however this can be an expensive product and hard to secure supply to a
large number of patients.
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
Discussion:
•
Digital PCR: A member noted they were not sure if there was a strong need for this in routine
laboratories. It was noted it may be useful in niche circumstances for immunosuppressed patients
with persistent COVID infection, or if there was a desire to precisely monitor viral load.
•
Host response molecular diagnostic: A member noted that these have been in development in
various settings for a long time, but have never been proven to have any clinical utility as immune
cell biomarkers are impossible to interpret clinical y and if not used in a clinical trial setting with
the key therapeutic included, they are not useful.
•
Saliva Direct: It noted this is currently in use in New Zealand and can be incorporated as
laboratories see fit and where there is a service need. It was noted by a member that if saliva
testing was felt to be important it should be decided by strategy rather than an assay.
•
Members requested further information when available on rapid testing, especially very rapid
PCR platforms that are up to date in their design and LAMP tests, and T and B cel immunity in
QuantiFERON assays for SARS-CoV-2.
•
The Chair raised a point fol owing interest in urine antibody testing.
1982
ACTION: The associated paper wil be shared with the group following the meeting for discussion in the
next meeting under horizon scanning.
ACT
4.0
Seroprevalence survey:
•
The Ministry noted they are establishing an Infection survey. Nasopharyngeal swab testing wil
proceed in early July fol owed by a seroprevalence survey for which the timeline is to be
confirmed. Design details are stil to be confirmed, however at this stage the survey wil be for
COVID-19 only.
•
CT TAG wil be updated by the Ministry as plans progress. Questions surrounding the survey wil
be brought to CT TAG in future meetings by SS&I.
ACTION: The Ministry wil provide an update of al surveillance work streams and E
INFORMATION SR surveilance at the
next meet including WGS surveil ance.
Discussion:
•
It was noted by the Ministry that the options for accessing passive surveil ance information with
change to mandates has required a reassessment of the current planning and the importance of
active surveil ance programmes is recognised for a r
OFFICIAL ange of respiratory pathogens including
SARS-CoV-2.
5.0
Serologic testing – Persistent infection
THE
•
A member updated progress on the
DRAFT Serological testing for SARS v3 document being
developed by the New Zealand Microbiology Network (NZMN) with minor amendments.
•
The difficulties of creating clear guidance on interpreting results were expressed, specifically for
antibody levels with result
UNDER s that sit in the middle range. Care needs to be given to developing
thresholds in this grey area when using this to decide on a patient’s eligibility for convalescent
plasma treatment or Evusheld.
•
It was noted further input from CT TAG members is needed into the document. This document wil
be brought back to the next CT TAG meeting.
•
Once the document is finalised the Ministry wil link into clinical and public guidance and wil link
with the COV
RELEASED ID-19 Therapeutics Technical Advisory Group (Therapeutics TAG).
Discussion:
•
It was noted this wil be a helpful document with a range of audiences.
•
A member commented on the limited supply of convalescent plasma and the need for it to be
rationalised and targeted to those who need it. The Therapeutics TAG may be the best option for
understanding the availability of this treatment. Due to the cross over with some of the work
between the two groups the action was raised to connect the chairs of the two meeting to discuss
further overlap.
•
The possibility of sharing minutes from the TAG meeting was discussed. This wil need to be
discussed between the chairs of the different groups to share this information.
ACTION: The Chair of CT TAG and Therapeutics TAG to discuss serologic testing and shared topics of
interest including sharing the minutes from the meetings.
ACTION: Members to update the paper and bring back to the next meeting.
6.0
Antibody Health Report on Serology and Immunity Testing
•
STA is currently working on translating the antibody memo into a Health Report for Minister
Verrall.
•
The first draft re
DRAFT Briefing Use of serology in COVID-19 CT TAG has been shared with the
agenda, this was shared as an initial draft for early comments and input from members. The
following questions were raised to the members:
o What are key messages that need to be brought through?
o What information would you advise should be included?
ACTION: Members were encouraged to send through their input to STA following the meeting.
ACTION: STA wil follow up in the next meeting with the final draft for review.
1982
Discussion
•
The members of CT TAG recommended that equity is considered consistently throughout the
ACT
document/report as opposed to a separate section at the end. This would require a change in the
Health Report/Memo template. STA wil feed this back to the Ministry for review.
•
A member noted serology testing could also be used for assessment of performance of Rapid
Antigen Tests (RATs) with either false positives or false negatives.
•
It was noted the use of serology for timing of boosters should be explored further. With additional
booster vaccines on the horizon. Timing of boosters and what testing markers could support this.
This could be of benefit for those who suffer negative side effects related to the vaccine.
•
It was noted some of the information from the COVID-19 Vaccine Technical Advisory Group (CV
INFORMATION
TAG) would be useful for CT TAG members and asked if it was possible to receive the minutes
from these minutes to better inform their decisions.
ACTION: STA to follow up on changing the content include equity considerations for each section.
ACTION: STA include in the HR consideration of utility of serology testing to inform timing of vaccine
boosters.
OFFICIAL
ACTION: The Chair of CT TAG to contact the Chair of CV TAG to discuss information sharing across
TAGs including minutes.
THE
7.0
Update on device applications and approvals (non-RAT)
•
The draft memo re
Memo Seeking approval to exempt POCT molecular from the Order DRAFT
was shared with the members in the updated agenda to seek input before going to the Director-
General for approval. Currently this is restricted to a few entities. This seeks to broaden the
UNDER
exemption to any device.
•
It was noted the need to open access to more sensitive and better performing tests particularly in
the at-risk groups and settings.
ACTION: Input from the CT TAG members by Friday, 20 May 2022 is requested.
8.0
RAT and PCR utility - HR Health Report
RELEASED
•
This item was noted and is stil in development. Members were requested to provide
recommendations to STA on key components that should be covered within the HR to support
future decision making and planning.
9.0
Next Steps/Decisions Pending
None noted
10.0 Any Other Business
None noted
11.0 Agenda Items for Next Meeting
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
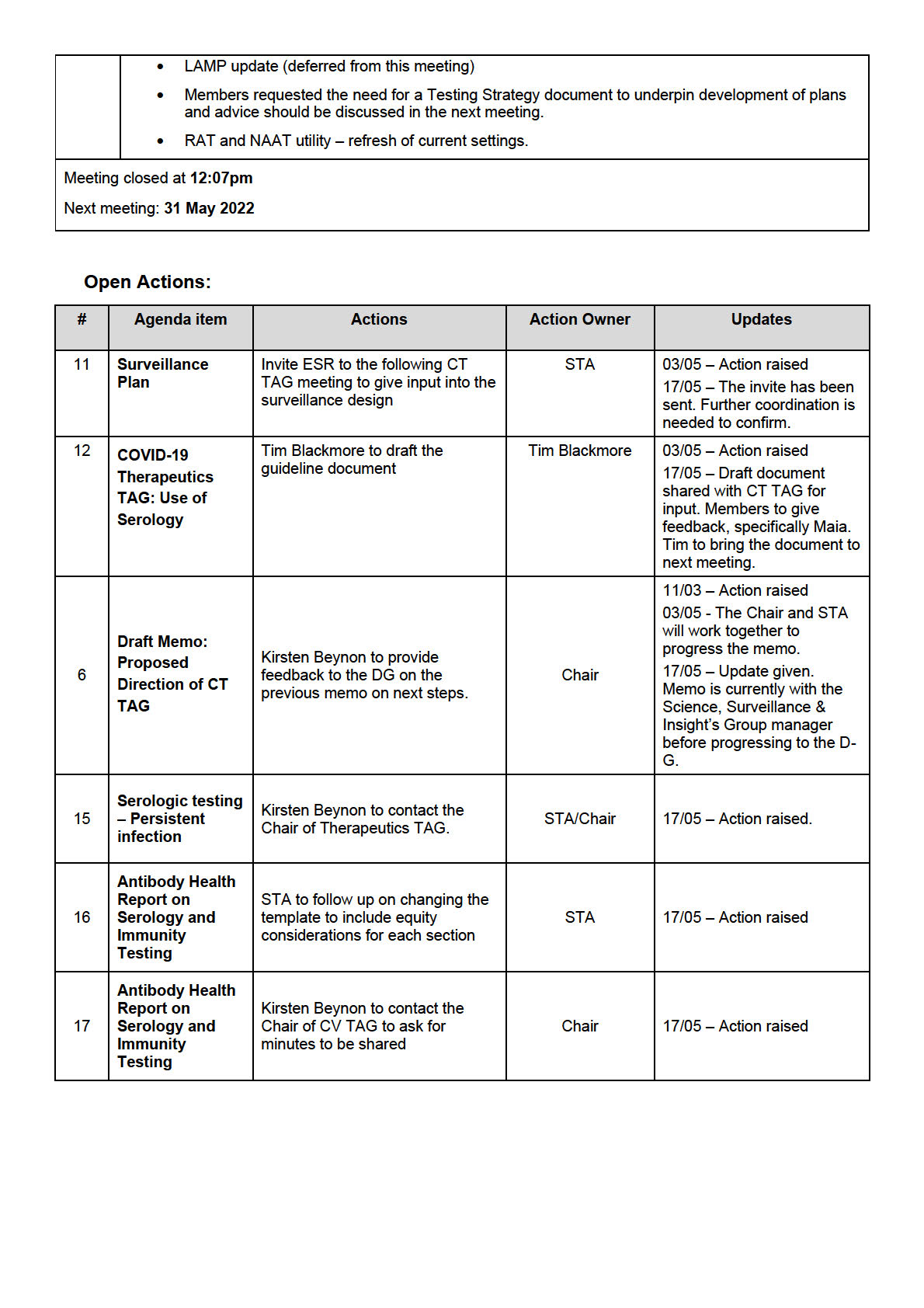
o members raised the increased importance of testing immunocompromised or people
with regular interactions with immunocompromised people
o the ability to detect reinfection requires accurate first detection. This depends on the
type of test (whether it was PCR or RAT, a self-test, or requested by a healthcare
professional)
o the appropriate advice wil be defined by the strategy and what it is trying to be
achieved (ie, different advice is required if the goal is to identify all cases, instruct them
to isolate, and prevent the spread of Omicron, or if the goal is to help those who need
treatment)
o it was acknowledged that the exact risk of reinfection is stil unknown, and depends on
vaccination status, whether the person is immunosuppressed and it is a persistent
infection or reinfection, or whether it may be a false positive.
• The TAG advised that the Ministry should continue to discourage people from testing too early
and unnecessarily. The message should continue to be to test only when it makes a difference,
and this should be consistent across other winter viruses.
1982
• CT TAG acknowledged that future variants are a concern which require flexibility and could see
higher rates of reinfection.
o Specific advice was requested on what testing regime to recommend to a G
ACT P in a
situation where a patient has plausible symptoms, if their second RAT is negative, and
whether it is enough to not note them as a case. It was raised that a second negative
test could be enough to consider the person negative, and that this has social and
economic impacts on the individual.
o Advice for symptomatic people should be to stay home. This is a pragmatic approach
and true for other viruses (Influenza, RSV).
o There was a call for integration of advice for COVID-19 and other winter respiratory
viruses to align.
INFORMATION
4.0
Update on Device Applications and Approvals (non-RAT)
A general update was presented on device applications and approvals.
• Work is underway to determine where the regulation of COVID-19 point of care tests will go
OFFICIAL
after transitioning to Health New Zealand and the Public Health Agency. An overview of RAT
and NAAT applications received was given.
THE
• Feedback was requested on the update, and the TAG were asked at what stage they want to be
made aware of applications for new devices.
Discussion:
• The pressure on evaluat
UNDER ing, approving and increasing capacity to deliver further Rapid Antigen
Tests is dropping due to the number already approved. It was noted that there is no opportunity
for quality assurance once a device is in the community, and no information on historically
approved devices and their detection or performance against new variants.
• A member updated the group on a meeting with New Zealand Microbiology Network (NZMN).
Concerns were raised on the currently approved molecular tests and what the scope is beyond
COVID-19.
RELEASED
o If these are removed from legislation it wil be difficult to reverse, as it would be difficult
to withdraw a publicly available test.
o The potential for scope creep for the testing to move beyond COVID-19 was noted.
There could also be health, safety and quality assurance issues.
ACTION: Periodic updates to be brought to CT TAG meetings about the progress of these applications,
with a particular focus on new emerging technologies and how they wil be used.
5.0
Infection and Seroprevalence Survey Overview
An update was provided on the oversight of seroprevalence and sampling. The Infection survey review
is underway. It was noted that there is an opportunity to collect reinfection data from this survey.
ACTION: Further documentation relating to this survey to be circulated to CT TAG.
6.0
Health Report Serology Antibody Testing - Immunity
• It was noted that the Report is a high-level update for the Minister. It has been circulated to CT
TAG members for feedback on key messages.
Discussion:
• Members noted some limitations of the document. It was noted that the Report does not allude
to the limitations of the quantification of testing, which can limit use when deciding if another
1982
vaccination dose is appropriate due to having no validated cut off and no correlation between
levels and protection. There is also not great reproducibility between tests.
• The Report could also mention new emerging technologies. There is no comment on T cell tests
ACT
and how things have changed in last month, and commercial surrogate viral assays are now
available which may move into diagnostics.
ACTION: Health Report wil be updated based on CT TAG feedback.
7.0
Airfinity update I Horizon scanning
• Information on rapid diagnostics and quantiFERON has been requested from Airfinity. This will
INFORMATION
be included in the next report.
• Any further topics of interest raised wil be passed on to Airfinity when raised by CT TAG
members.
ACTION: Rapid diagnostics and QuantiFERON deep dives to be reported to CT TAG when available.
OFFICIAL
8.0
Testing Strategy
THE
This item was not discussed.
9.0
Next Steps/Decisions Pending
None noted.
UNDER
Any Other Business
10.0
None noted.
11.0
Agenda Items for Next Meeting
RELEASED
Meeting closed at
11:59 pm Next meeting:
14 June 2022
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
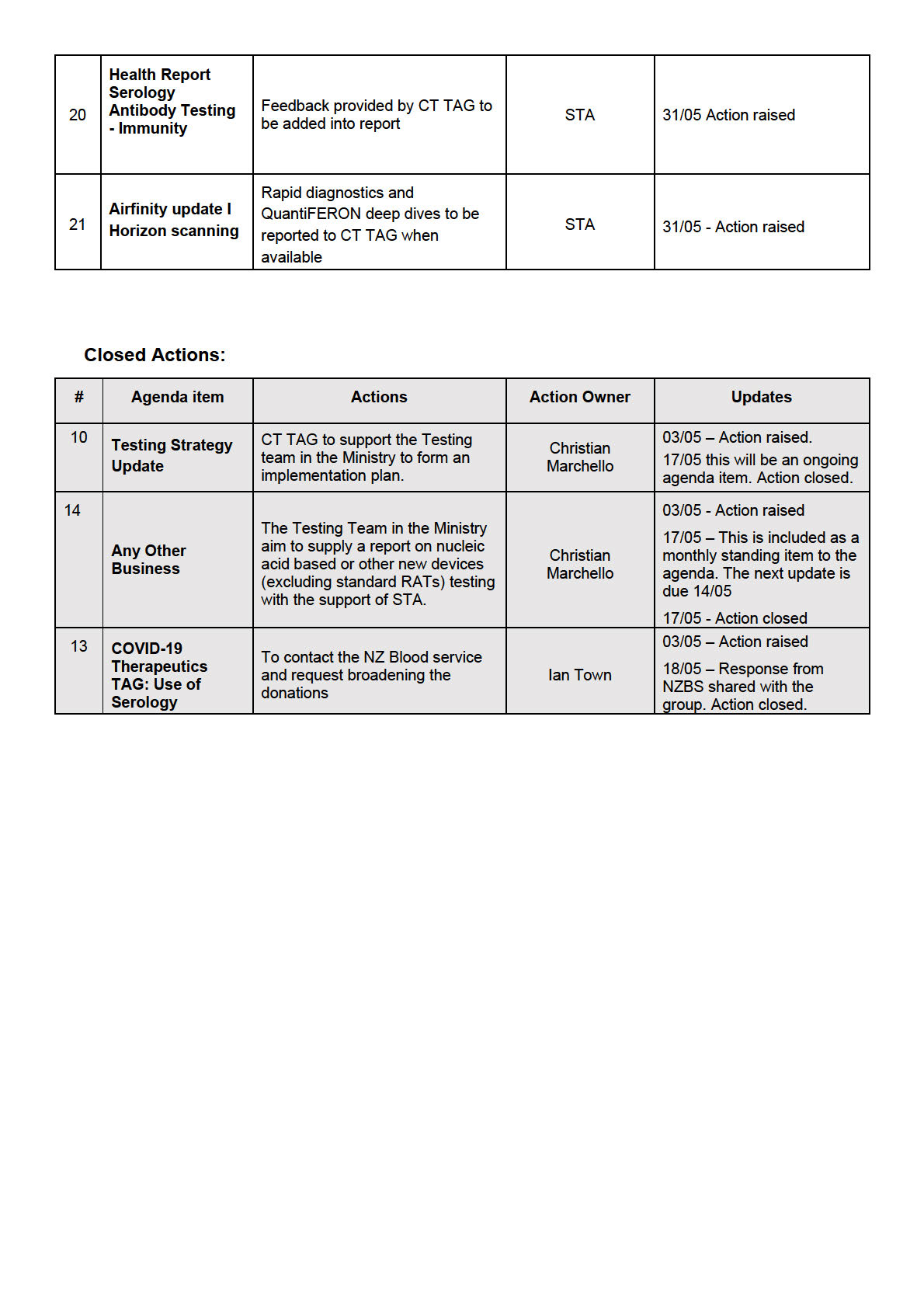
o for early detection on ILI activity, seasonality, the start and end of
traditional influenza season, and to provide historic rates for annual
comparisons
o to gain information on which viruses are circulating and where
• This information wil be used long term to inform vaccine design and policy.
• It was noted that the sentinel surveil ance wil use the ILI definition (fever and
cough), which wil only capture a small subset of COVID-19 cases.
• While system development is occurring throughout the season, the weekly ILI
dashboard has been stood up. This includes 54 sentinel GP practices utilising the
current existing systems (with an embedded HealthLink form).
Discussion:
• Incorporation of existing laboratory respiratory viral testing information into the ILI
1982
surveil ance reporting.
• It was recommended by a member CTCs may be a source to increase ILI update
ACT
and do panel testing on some of these samples.
• There is an aim for ESR to get data through the result depository moving forward.
• A member questioned whether Shivers data would be included. Shivers practices
are using an RE case definition rather than ILI definition, but noted that there is rich
data available there.
• A member emphasised the importance of real time information on circulating
viruses at a local level to support clinicians. It was observed in the past there was a
delay in updates from the ILI sentinel surveil ance. Weekly information is now being
INFORMATION
uploaded.
• The TAG highlighted the importance of push notifications processes to updating
relevant groups.
• It was also noted that it comes down to which testing panels are run (triplex or
multiplex). Push notifications to convey this would be very useful. CT TAG wil look
OFFICIAL
at some options for a push notification system at both a national and regional level.
• Another member noted that influenza and RSV activity in Australia should also be
incorporated. Often a couple of weeks after Australia sees an upsurge in either
THE
influenza or RSV we also see an upsurge in case numbers in New Zealand. ESR
advised this is occurring.
• A member noted there is a combination of datasets being gathered and encourage
ESR to lead integrat
UNDER ion of this.
4.0 Airfinity update I Horizon scanning
• Airfinity scanning on QUANTiferon will be circulated and feedback is to be given by
CT TAG at the next meeting. A member commented that the review does not use
the same platform as QuantiFERON so it is a more complicated Elisa method, and
the labs
RELEASED cannot jump in and do this easily for verification.
• There is ongoing engagement with the Doherty Institute as part of horizon
scanning.
• It was noted that there are a few quick to market RAT saliva tests which have been
released to market in some jurisdictions.
• Slides and paper from Airfinity to be sent to TAG members before next meeting.
Discussion:
GPs for urine antibody paper
• A member noted issues with the study design and method description being
unclear and not wel defined. This was deemed not relevant for a NZ context.
Recommend no diagnostic utility for this method at this time.
• Another member noted that a wide range of results for urine is unsurprising as
urine samples can be very concentrated or diluted depending on hydration levels
and timing of collection, and therefore it cannot be used quantitively.
5.0 Persistent infection
• An updated version of the persistent infection and serology testing was received
from NZMN.
Discussion:
1982
• The document was formal y endorsed by CT TAG. It was noted that as more
information comes to light, the situation is likely to change.
• A discussion occurred on distribution and whether it should be made publicly
ACT
available through the NZMN or Ministry of Health websites. It was queried whether
it should be formal y sent to the group who worked on the persistent infection
guidelines, as it could potential y be linked.
• It was noted that this was written more for the benefit of the TAG than for the
general public, and there was not considered to be a wide readership.
o It was agreed that this would be most appropriate place for clinicians looking
after patients who are immunosuppressed likely to have persistent COVID-
19 infection.
INFORMATION
o The idea of using push notifications was also raised as a way of updating
clinicians on new information made available as it is not currently clear
where persistent infection guidance goes, as this is not a national clinical
care formal document. Mostly because its all-expert opinion with some data
behind it.
OFFICIAL
ACTION: Discuss with Therapeutics TAG and STA to consider incorporation into existing
Clinical Guidance from a MOH perspective.
THE
6.0 Update on Device Applications and Approvals (non-RAT)
• s 9(2)(f)(iv)
UNDER
• CT TAG were asked for comments relating to self-use NAATs, recognising the
public can choose to purchase it similar to purchasing RATs privately.
Discussion:
• A member asked about the funding criteria/process.
• Concerns were raised regarding the lack of clarity of quality assurance processes,
RELEASED
and the need to keep up to date with new variants.
• It was noted that previous discussion has occurred with the NZMN about the point
of care nucleic acid tests, with feedback given on the potential issues with health
and safety, quality assurance and data governance while fol owing privacy
guidelines.
o It was noted that RATs were made available rapidly before mechanisms to
capture results were available. This was needed due to qPCR testing
being overwhelmed.
o The context in which NAAT’s are used need to be carefully considered
before being made widely available.
• Another member mentioned that it comes down to accessibility of tests and
reporting requirements need to be addressed before release to the market. If this
can be done, then some parts of the country with accessibility issues are likely to
benefit from NAAT tests.
o A member also advised if these tests were run by point of care teams with
training and reporting, then these issues might be mitigated but opening
sale to anyone has many potential issues with regulation.
•
The importance of clear with the public messaging on utility of tests and potential
downsides of each. It was noted there is a general lack of public understanding
1982
regarding sensitivity for RATs and NAATs can detect genetic material after a person
is no longer infectious. Businesses also need to be aware of other respiratory
il nesses outside of COVID-19. The TAG supports the message of if you are u
ACT nwel
stay home; seek health professional support if you have underlying conditions or
your symptoms worsen irrespective of COVID-19 testing results.
• s 9(2)(f)(iv)
7.0 Proposed direction of CT TAG memo
•
The Ministry leads met with the Director-General last week where the importance of
the TAG was acknowledged.
•
An update was provided on this memo detailing the future direction of CT TAG. This
INFORMATION
is stil in progress.
Discussion:
• A member questioned whether the purpose of this group is going to mainly be
focused on the technical aspects of new technology and clinical scenarios. The
member noted the lack of external advisory on how tests are used in surveil ance and
other public health responses and how CT TA
OFFICIAL G can assist with this.
• The TAG noted a need for clearer terms of reference and role or advice on
surveil ance.
THE
• The Chair and STA wil finalise memo the memo and bring it back to CT TAG.
ACTION: Finalise memo and bring back to CT TAG.
8.0 Engagement with Therapeutics TAG
• The Chair met with
UNDER the Chair of the Therapeutics TAG to discuss how issues
pertaining to both groups could be discussed.
• Moving forward minutes from CT TAG and Therapeutics TAG wil be shared after
each meeting. STA team wil coordinate this.
9.0 Reinfection Update
• Interim advice wil be released; this wil be under regular review.
RELEASED
• The updated guidance was overviewed by the team for the Chair.
Discussion:
• A member noted that the guidelines are very complicated and need to be tailored
for each target audience. The key message to emphasise is that if you are unwel ,
stay home.
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED

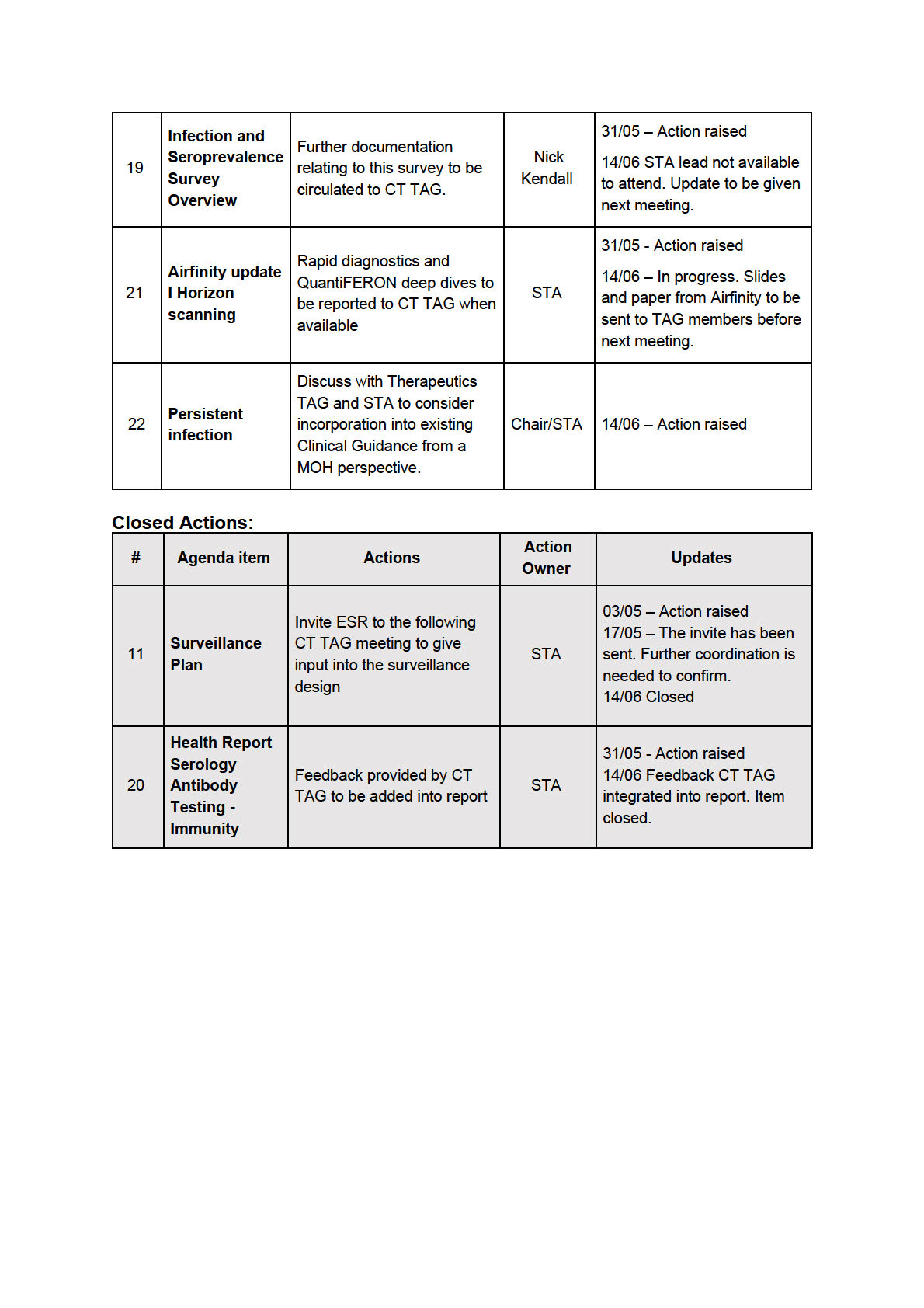
Document 5
MINUTES: COVID-19 Testing Technical Advisory Group
Date:
12 July 2022
Time:
11:00 am to 12:00 pm
1982
s 9(2)(k)
ACT
Location:
Chair:
Kirsten Beynon
Members:
David Murdoch, Maia Brewerton, Patricia Priest, Pisila Fanolua, Susan
INFORMATION
Morpeth, Tim Blackmore
Ministry of Health
Brittany Il ingworth, Brooke Hollingshead, Christian Marchello, Imogen
Attendees:
Roth, Mark Ayson
Guests:
Fiona Cal aghan, Nick Kendall
OFFICIAL
Apologies:
Christian Marchello, David Murdoch, Imogen Roth, Pisila Fanolua,
Susan Morpeth
THE
1.0 Welcome and Previous Minutes
Kirsten Beynon welcomed all Members and Attendees in her capacity as Chair of the COVID-
UNDER
19 Testing Technical Advisory Group (CT TAG).
Minutes of the last meeting (14 June 2022) were accepted.
2.0 Update on Open Actions
N/A
RELEASED
3.0 Seroprevalence Survey Update: Questions about SARS-CoV-2 antibody testing used in
Sero surveil ance
An update was provided on the seroprevalence survey. Key questions that are in need of
advice include:
-
Q1. How do we take into account waning Ab levels over time especially anti-N as it
seems to wane faster than anti-S?
- Q2. What is your advice regarding a single (cross-sectional) Ab level test vs. serial Ab
levels?
* Note, there is potential to collect data from: multiple cross-sectional population samples (eg,
weekly for 6-months); and an additional sub-group recruited for repeat testing (eg, monthly for
6-months).
- Q3. Where would you recommend, we access blood samples (eg, random pop
sample, residual, or blood donors?)
Discussion:
Q1 feedback
1982
• An epidemiological serosurvey wil provide a snapshot of the past and is not suited for
purpose in the current fast-paced environment of emerging SARS-CoV-2 Omic
ACT ron
variants. Members questioned the value of a seroprevalence survey that included anti-
nucleocapsid antibody testing for the following reasons;
o we already know that many have had COVID-19
o it wil underestimate cases
o it wil not be possible to get a ful picture of everyone who's had an infection
o it wil not provide insight into immunity against future strains.
• Serosurveys need to be in context of what virus variants are currently of concern and
what variants are coming (ie, how a population previously infected with Omicron are
INFORMATION
responding to latest (or future) variants).
• With COVID-19 and emerging variants the timing of a serosurvey is critical to inform
immunity and decision making; the Ministry should consider waiting until the current
wave has passed.
• The purpose of a seroprevalence survey is to use the most appropriate test as a
OFFICIAL
surrogate for immunity and for informing vaccination decisions. Standard commercial
anti-nucleocapsid antibody testing wil not provide this information.
• It is recommended by TAG mem
THE bers that pseudo-virus neutralisation assays are
considered for any seroprevalence study undertaken. They are relatively simple to
perform, are updated regularly and a laboratory could implement this if needed. Also,
knowing whether antibodies can neutralise (circulating or new) variants is more useful
than determining if people have had an infection.
UNDER
Q2 feedback
• The overal purpose of the surveil ance should inform which assays are selected and
the sampling design.
• The TAG asked for clarity on how this wil feed into future decision making for public
health.
RELEASED
Q3 feedback
• The Ministry needs to be clear on the purpose of the survey before determining
sampling design and access to samples/participants.
• Cross sectional sampling: the timing of the serum sample being taken relative to
infection and/or vaccination and within a relatively short timeframe is important. Also,
TAG members suggested that it should be asked what would be gained from a public
health perspective from the investment.
•
A priority objective should be to identify who are our most vulnerable populations or
individuals.
Key Feedback from CT TAG Members
o
Pseudo-virus neutralisation assays are investigated for use in the
seroprevalence survey as they are the most suitable test to give information
about population immunity.
o
The Ministry should consider the value of undertaking a cross-sectional 1982
seroprevalence survey at a single point of time from a public health perspective.
o
Sampling and study design needs to be dependent on the overal purpose of the
seroprevalence survey.
ACT
o
Identify the most vulnerable populations.
o
CT TAG members requested that papers submitted have a clear purpose and
executive summary for ease of reading.
ACTION: Feedback to be sent to ISK surveil ance team.
4.0 Whole genome sequencing (WGS) update
Dr Fiona Cal aghan presented to CT TAG an update on wastewater (WW) and WGS
INFORMATION
surveil ance programs in place.
Discussion:
Members noted that there would be regional differences and inconsistencies in sampling. For
example, labs in Wel ington are using POCT devices as their primary testing method so
numbers of samples available for WGS testing is lower.
OFFICIAL
It was also noted that Auckland WGS testing is disproportionately low based on population and
vulnerable population profile compared to other regions.
THE
•
It was noted work with ESR and testing labs is in progress to achieve more consistent
and representative sampling by region.
Members observed and encouraged that longer term cost/benefit analysis should be
undertaken as to the value of
UNDER focussing on only COVID-19 testing.
•
It was noted more complete surveil ance data over the next wave wil be of value to
inform future surveil ance priorities and design.
A concern was raised regarding the value of WGS for assessing reinfections:
Members noted:
RELEASED
•
differentiation by WGS at an individual level to assess persistent- versus re-infection
would be of benefit for a small population (the immunocompromised)
•
as WGS is focussed on population surveil ance, the systems are not wel set up for
reporting individuals' information to clinicians or the patient. WGS results need to be in
the patient record to be of value for clinicians managing patient's care including access
to previous RAT results
• as per current protocols, for patients who wil benefit from targeted therapies, WGS
can be of value irrespective of first, second or third infection.
It was noted that reinfections are now being captured and rates wil be monitored against WGS
population surveil ance from WW, border, hospital, and community.
5.0 Airfinity update I Horizon scanning
STA presented an update, which included key updates on developing novel diagnostics using
saliva with a recent study looking at a lol ipop RAT. Several papers looked at gargling as a way
to collect a specimen. There were also additional papers about QuantiFERON but stil lacking
1982
a link to clinical response. Final y, it was shared that the Ministry are meeting with the Doherty
institute regularly to share experiences and discuss testing modalities. Multiplex rapid antigen
tests are not going ahead in Australia, while multiplex PCR is being used in selected settings.
ACT
Of interest, they have had success in undertaking sequencing using the paper strips inside
used rapid antigen test devices and are currently working to operationalise this. Deep
engagement with labs is required for this to be successful.
6.0 Ministry of Health update
- To be sent by email
ACTION: STA to share an update via email outlining the new Ministry structures
INFORMATION
7.1 Any Other Business
The Surveil ance Strategy, Testing Plan, Reinfection Guidance and Infection survey were
shared with CT TAG for noting so that they are aware of the current versions and status of
these.
OFFICIAL
Work is being undertaken on updating the Testing A3 and Clinical Guidance. This wil be
shared with CT TAG for comment once the new format is agreed and content updated.
THE
ACTION: Add to agenda template ‘AOB’ time to discuss new issues as raised by members
7.5 Rapid regulatory review underway for POCT devices approvals
Members were informed this review was underway.
UNDER
Members requested regular updates on the ongoing post market monitoring of devices that are
used in the open market. Particularly performance against emerging Omicron and other
variants.
ACTION: Process to be established by STA for regular monitoring or recalling approved RATS
and monitoring performance against new variants.
RELEASED
7.6 Cross government workshop on testing innovation
Members were informed this is underway, as part of a wider review of the end-to-end process
of the innovation pathway.
Members noted ongoing interest in this work and implications for all medical devices.
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
•
There was a spike in demand for inhaled budesonide in March. AstraZeneca and Pharmac are
monitoring what this means for stock holdings. Current stock holdings are between 2-7 months.
Pharmac is interested in feedback from the group on how useful this is for treating COVID-19.
•
Remdesivir: following a shipment last week there is an increase in stock from what was
communicated previously.
•
For baricitinib, both 4mg and 2mg remain available. Pharmac are working with the supplier to
establish longer term supply.
•
Tocilizumab: Pharmac are working with the supplier to establish the next delivery.
•
Pharmac has concluded the initial discussions with Novartis (ensovibep) and GSK (sotrovimab)
about COVID-19 treatments and is currently waiting for the clinical information from the supplier
before bringing to the advisory group for assessment and recommendations.
Medsafe Update
•
The Medsafe assessment for molnupiravir is complete. This wil be referred to the Medicine
Assessment Advisory Committee (MAAC) on 12 April 2022 for advice.
1982
STA Update
•
Airfinity provided information about a decrease in neutralisation against omicron sub
ACT -variants BA.1
and BA.2. for both Evusheld and sotrovimab.
Discussion:
The Chair noted it would be important to consider the role of Evusheld and sotrovimab by the time they
become available in New Zealand.
3.0
Equity Considerations
INFORMATION
•
The community space has been busy with the roll-out of Paxlovid. Webinars, readings, and peer
groups are being hosted to educate the community on the best way of getting Paxlovid to those
who need it. The regional roll-out is being incorporated into local health pathways.
•
It was noted there is variation of practice in the community specifical y around prescribing
budesonide. There is concern about this, particularly with the rol -out of further medications
OFFICIAL
especially in improving communication to and by community providers. Noting there was a need to
simplify the drug guidance provided to community providers.
THE
Discussion:
The advisory group acknowledged these challenges and noted that there was significant work going into
producing infographics and easier-to-follow guidance.
UNDER
4.0
Paxlovid roll-out
•
Paxlovid became available to prescribe on 5 April.
•
The access criteria for Paxlovid was published on the Pharmac website on 31 March 2022. This
links back to the Ministry webpage for who is at high risk of severe outcomes with COVID-19.
•
Health Pathways have published a localised list of pharmacies where Paxlovid is available.
RELEASED
•
A supporting resource was produced by He Ako Hiringa that provides guidance for clinicians
regarding drug interactions.
•
Health Navigator has worked with He Ako Hiringa to make sure there is a consumer information
pamphlet handed out when dispensing. This has been translated into 6 more languages.
•
There is going to be a combined webinar on 13 April with the Ministry, The Col ege of General
Practitioners, and Pharmac. This will largely focus on Paxlovid prescribing and dispensing.
• A second shipment of Paxlovid has arrived meaning there are more courses available than
original y anticipated. Pharmac is not expecting any future shipments of Paxlovid before July 2022.
• Since becoming available there has been slightly less courses of Paxlovid dispensed than
expected.
• Of the original shipment 15 percent has been kept as an essential reserve. A further 15 percent
has been reserved for the localities. This al ows pharmacy portfolio managers to approve further
orders from the reserve stock if needed.
• ProPharma is the community wholesaler and contracted provider to distribute stock. Every
pharmacy that has been identified as a participating pharmacy is now able to order stock.
ProPharma does not usual y distribute to hospitals.
• Due to low stock the initial decision was made to not supply Paxlovid to hospitals. The aim of
Paxlovid is to avoid the need for people to go to hospital. Those that meet the access criteria are
likely to require ongoing monitoring through primary care. The limitations of ProPharma distribution
1982
and data collection in hospitals also aided this decision.
• The monitoring tool that has been developed relies on NZEPS data for what has been dispensed.
ACT
This is not currently used in hospitals. Consideration needs to be given for maintaining oversight.
• Supplying to hospitals aims to make Paxlovid available to patients who are currently taking the
medication and admitted. Additionally, patients may be diagnosed with COVID in hospital and
Paxlovid may be the best course of treatment. There also needs to be a supply in EDs for people
visiting ED that may not otherwise interact with the health sector.
• It was noted that rural hospitals have been able to access stock provided through local
pharmacies.
INFORMATION
• The Ministry’s data and digital team have created a dashboard that calculates:
o How much stock is available at the wholesale level
o How much is distributed to pharmacies
o How much DHBs have available
OFFICIAL
This tool also captures the demographics of patients being prescribed Paxlovid. The aim is to
ensure the prescribing of Paxlovid is equitable and to help inform possible changes needed to the
THE
access criteria if stock becomes limited.
• It was noted the access criteria now includes high-risk individuals who have symptoms of COVID-
19 but aren’t a household contact and don’t have access to a PCR test within the therapeutic
window.
UNDER
Discussion:
A member noted that as NZ moves into winter, there wil be a greater prevalence of other viruses with
similar symptoms. As there is limited supply of Paxlovid, rapid PCR tests need to prioritised going forward.
This feeds back into the testing strategy and how the vulnerable population are identified.
STA have been in discussion with the Chair of COVID-19 Testing Technical Advisory Group in regard to
RELEASED
timely access to PCR tests. A position statement is currently in progress for this.
The group agreed some attention may be needed for publicising the availability of Paxlovid to the public,
so they are aware this is a treatment option.
Proposed changes to Paxlovid access criteria:
• Currently, being incompletely vaccinated is only a risk factor when combined with age in the 50-65
year old group. It is proposed that vaccination status should be an independent risk factor for this
age strata.
• The proposed changes are as follows:
o That age over 65 years accounts for two risk factor points
o That the age strata of 50-65 years counts as one risk factor point regardless of vaccination
status
• There is current work towards creating one set of access criteria for al antivirals currently
available. The aim is to set a broader threshold for considering a patient for one or more options of
early treatment. Within this access criteria, there needs to be a way to prioritise the different
treatment options.
Discussion:
1982
Pharmac was happy to receive and consider the proposed changes.
5.0
Horizon scanning
ACT
Molnupiravir
• It was noted the need to be clear about the role of this drug in the community rol -out of anti-virals.
Noting it should not be prioritised over the other treatments available. The access criteria should
account for this within the gradient/risk matrix for treatment proposed in the guideline update.
• Medsafe noted the availability of molnupiravir depends on advice from MAAC. The time frame for
this can vary.
INFORMATION
6.0
Guideline Updates
• Last guideline update was on 1 April with the addition of Paxlovid as an option for early treatment.
With this addition, remdesivir has moved down the list in order of treatment. An infographic was
included to give an overview of the early treatment of COVID.
• To simplify the guidance further, it is proposed to have a single access criterion. This wil include a
OFFICIAL
risk matrix for guidance on how to choose between the different treatment options. The risk matrix
is formatted in a similar way to the cardiovascular risk matrix.
THE
• The risk matrix creates an order of preference represented by red, orange, and green. Those in
the ‘red’ group require the highest priority of treatment (i.e., Paxlovid). Patients who meet the
access criteria but are in the lower end of the threshold could be considered for other options if
Paxlovid is not available and remdesivir isn’t suitable.
UNDER
• Anyone who is severely immunocompromised is automatical y considered to be ‘red’. This criterion
has been removed from the graph to further simplify this.
Discussion:
Phamac noted the system in place stil requires bul et point lists to be published, however it is possible to
incorporate the graphical risk tables.
RELEASED
It was noted by a member the difficulty in reducing complexity. Real data and evidence should be used to
refine and formalise the advice for each colour group. This further brings up the need to define what is
meant by ‘fully vaccinated’ and identify who is most at risk.
7.0
Any Other Business
8.0
Agenda Items for Next Meeting
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
population. The proposed changes to the access criteria and risk matrix increase the
eligibility to approximately 1.5 percent of the population (around 75,000 people).
• Pharmac is currently considering updating the risk matrix and access criteria to extend this to
2 percent by extending the access criteria to include the fol owing:
o other ethnicities over the age of 65 with one co-morbidity
o Māori and Pacific peoples over the age of 25
Pinnacle model ed an additional 0.5 percent of the population becoming eligible to bring the
total eligible population to 2 percent. This would increase the amount of eligible people to
approximately 100,000, in line with the amount of Paxlovid courses for treatment available.
Discussion:
• There was discussion about what the threshold risk of hospitalisation should be, with members
commenting this could be in the range of 5-10%. There are limitations to the data on risk of
hospitalisation, particularly for comorbidities.
• A member asked if it was possible before publishing to analyse the risk for each box repres
1982 ented,
to confirm if the highest risk groups are being accurately identified. Pharmac noted they are using
data from British Colombia (BC) to analyse this. This BC data was col ected between December
ACT
2021 and January 2022. Pharmac acknowledges the data from BC does not represent the
ethnicity represented in New Zealand.
• Pharmac acknowledge the data col ected from the Auckland hospitals. This data shows the strong
risk associated with age and the need for this to be weighted appropriately in the risk matrix. The
data also shows vaccination status as the next strongest driver for risk. From this data, Māori and
Pacific peoples present the highest risk for ethnicity groups.
• It was suggested by several members to include percentages in the risk matrix if there is sufficient
information available surrounding co-morbidities in New Zealand.
INFORMATION
• A member noted the low number of courses prescribed for Paxlovid and encouraged efforts to
extend the access criteria.
• A member raised concern over the possibility that molnupiravir would become the preferred
medication to prescribe over Paxlovid due to the ease to prescribe.
• A member noted the possibility to further stratify the age categories to provide a more nuanced
OFFICIAL
approach from those aged 65.
• Pharmac welcomes feedback and acknowledges there wil be further input considered going
forward.
THE
ACTION: The Ministry will look into data which could better inform the percent risk of hospitalisation.
Medsafe Update
• Molnupiravir (Lagevrio): no
UNDER w approved under s23 (provisional approval) on 14 April 2022.
• Evusheld: Medsafe is expecting an application from AstraZeneca middle of the week beginning 25
April.
• Actemra (tocilizumab): In relation to the Changed Medicine Notification (CMN) application to
extend indications to include COVID treatment, the evaluation has been completed and a decision
on approval is imminent
RELEASED
STA Update
• STA brought the members attention to the Airfinity report shared with the minutes, and in particular
the information on antivirals being administered intranasally with studies on hydroxiclorum and
remdesivir.
• STA is expecting a further update on the infographics for the following meeting
3.0
Equity Considerations
•
There is ongoing concern for the prescribing of Paxlovid mainly with patients recognising and
presenting within the first five days of infection. It was suggested stronger public messaging
surrounding available treatments be prioritised.
•
The number of patients presenting is also significantly lower than that previously. This factor adds
to the lower-than-expected numbers of Paxlovid being dispensed.
Discussion:
•
Pharmac acknowledged the efforts of Pinnacle at identifying at-risk groups and making direct
contact with information surrounding treatment options. It was suggested this level of personalised
care be taken into consideration in the primary care sector.
•
A member questioned the practicality of GPs pre-emptively reaching out to high-risk patients
understanding the restrictions in resources.
1982
•
A proposal for oseltamivir funding for influenza over the coming winter viral season was discussed,
which Pharmac acknowledged as an important concept to consider.
ACT
ACTION: STA to relay information to the comms team and suggest a marketing campaign for therapeutic
options for high-risk individuals.
4.0
Primary Care Update
•
Currently 689 courses of Paxlovid have been prescribed. This is lower than anticipated.
•
Molnupiravir communications update: Health Navigator are planning to release a plain language
information sheet; the New Zealand Formulary (NZF) have released a drug monograph; and He
INFORMATION
Ako Hiringa are creating a resource to be available with the roll-out of molnupiravir.
•
There is currently no webinar planned for this release of molnupiravir. In comparison to Paxlovid
there is less complexity involved in prescribing.
•
The slides presented wil be circulated following the meeting. Feedback from the group is
OFFICIAL
welcome.
Discussion:
THE
•
A member raised concern regarding the risk of pregnancy, as it was noted that the baseline risk for
severe outcomes of COVID-19 would not meet the threshold risk criteria.
•
It was noted that Paxlovid should be prioritised over molnupiravir. Pharmac is currently in
discussion about including t
UNDER his in the guidelines.
5.0
Immunosuppressed
Perspectives on serology use
•
Currently it is known that the serological tests can vary by about 30 percent between assays. Its
best application is in recognising those with very high or very low immunity. In most cases it is
RELEASED
being used to identify those who are severely immunocompromised, who have not clearly
mounted an immune response to vaccination.
•
In some situations, serological testing is being used to assess immune responses in people for the
eligibility of ongoing antiviral therapy and modulators.
•
Rapid Antigen Tests are known to be 30 percent less sensitive to the Omicron variant than to
Delta. Serology testing can play a role in identifying what portion of the population have been
exposed to COVID-19 and who is immune.
• In some cases, it helps people assess their own risk avoidance. It can give peace of mind to
patients that receive a high immune response, especially those with other medical conditions.
Discussion:
• A member noted that serological testing could be an important tool in identifying those who are
severely immunocompromised for the purpose of prescribing Evusheld.
• Specialists should be looking after patients with diagnostic tests to help guide advice. The Ministry
in interested in conducting a seroprevalence survey in order to understand who in NZ has been
exposed but are not currently looking to base therapeutic advice on this.
Northern region guideline on Persistent SARS-CoV-2 Infection
• Concern was raised on the efficacy of antiviral monotherapy. The member encouraged increasing
communication, information, and availability of Evusheld in NZ and stressed the need for an
effective antibody therapy.
1982
• Convalescent plasma treatment has been used on occasion with the support of NZ Blood Service.
This has been effective anecdotally particularly for eliminating persistent infection in profoundly
immunosuppressed patients. The NZ Blood service is currently reserving supply for
ACT a REMAP-
CAP trial to clarify the usefulness of the treatment.
• The guideline deals with the persistent infected group and caring for the severely
immunocompromised. This falls outside of the scope of the Hospital Guideline. Discussion on
turning this into a formal guideline is welcomed.
Discussion:
• A member added support noting there are currently very sick people experiencing long hospital
INFORMATION
stays and again stressed the need for an objective monoclonal antiviral as pre-exposure
prophylaxis for COVID-19.
6.0
STA Information Request
• STA wish to highlight the recent information available for risk based on time since vaccination. The
OFFICIAL
latest information stems from three large studies which have been shared with the group
• The primary goal of vaccination for SARS-CoV-2 remains protection against severe disease,
THE
hospitalisation, and death. Efficacy against protection against infection is known to wane after a
primary vaccination course.
• SARS-CoV-2 therapeutics may be used in some clinical situations where patients with COVID-19
are more at risk of hospitalisation and death
UNDER
• There is limited data looking at the long-term (more than six months) effectiveness of vaccination
in preventing hospitalisation and death
• There is no data looking at long-term vaccine effectiveness in preventing hospitalisation and death
across Omicron-dominant time periods, nor data from booster doses fol owing a primary
vaccination course
• There is no data looking at important intersecting factors for the New Zealand setting, in particular
Māori and Pac
RELEASED ific peoples, who are known to have poorer outcomes as a result of SARS-CoV-2
infection
• Current data indicates that eight months after the first dose, two doses of the Pfizer vaccine
provides ~90% protection against hospitalisation and ~90% against death
• Current data indicates that protection against hospitalisation death from two doses of the
AstraZeneca vaccine is not sustained to the same level, particularly in elderly and clinically
vulnerable populations
Discussion:
• A member noted recent literature showing a significant drop in vaccine effectiveness over time
with the Omicron variant.
• A member commented on personal clinical observation of people with two doses six months prior,
presenting similar to those who are unvaccinated.
7.0
Guideline Updates
• The next guideline update is planned for 29 April 2022.
• The guideline may require further updates to include molnupiravir as it is likely this will become
available in hospitals. There is the possibility for a delay in supply to hospitals similar to Paxlovid
however this is uncertain as Onelink has been established.
• The next guideline would likely include the updated risk matrix from Pharmac.
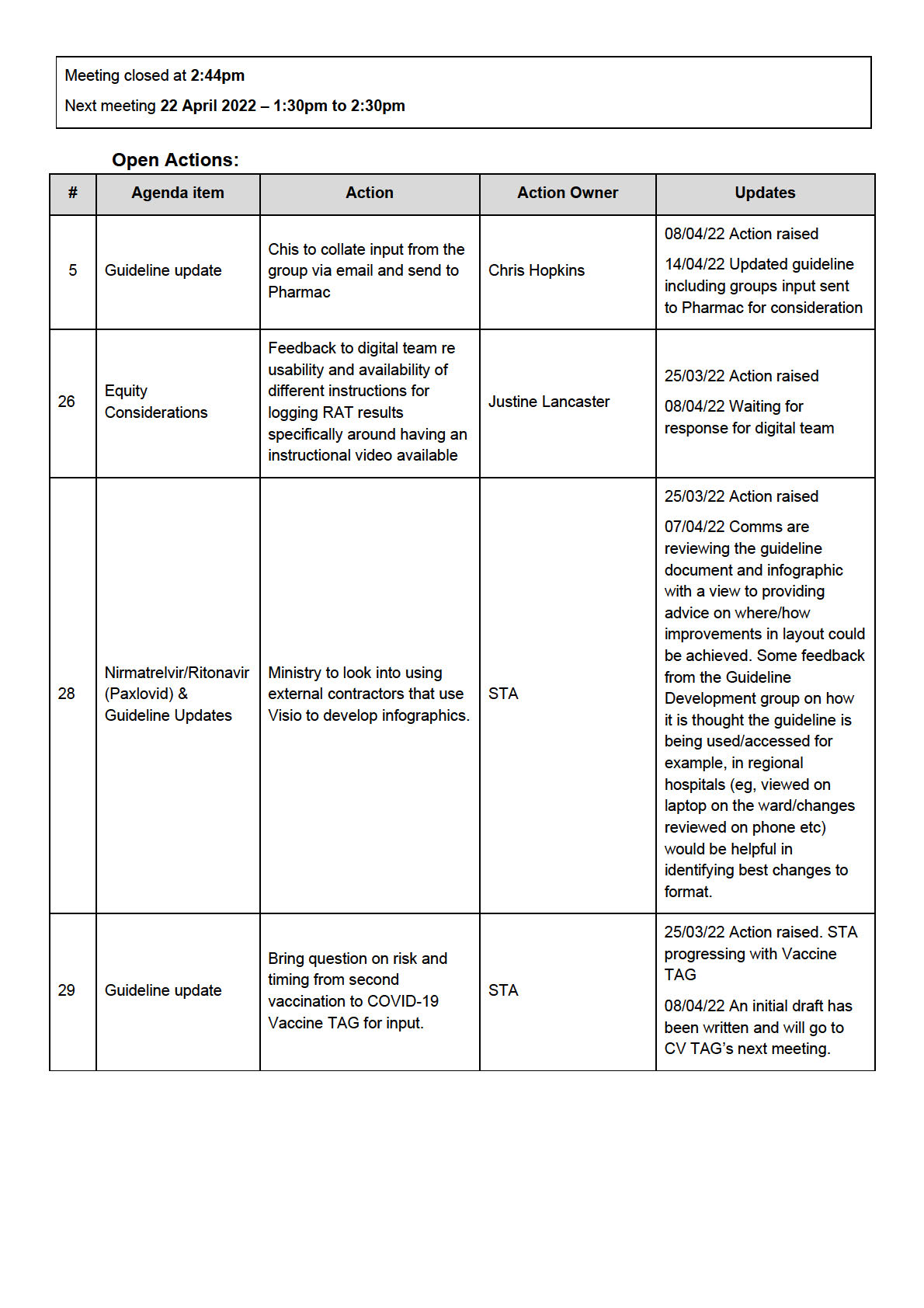
ACTION: Pharmac to communicate the availability of molnupiravir in hospitals before the publication of the
guideline update scheduled 29 April 2022.
1982
8.0
Next Steps/Decisions Pending
It was noted by the Chair to further include the percentage risk into the risk matrix. This is to be led by the
ACT
Ministry. There was discussion on inviting an expert in epidemiology for the meeting 6 May 2022 to gather
knowledge on the work being done in this area.
7.0
Any Other Business
None noted
8.0
Agenda Items for Next Meeting
INFORMATION
None noted
Meeting closed at
2:40pm Next meeting
06 May 2022 – 1:30pm to 2:40pm
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
o There are no updates to the usability of systems, however there is awareness that the rate
of self-reporting of RATs has gone down.
o It was noted that there may be other factors contributing to the reduced rates of self-
reporting of RATs than just the data loading and/or technical issues. This could be due to
attitudinal issues within the population towards COVID-19 and COVID-19 testing more
generally, and therefore requires broader communication strategies, particularly for winter
planning.
o This item is now closed.
• Infographics to assist with the current Paxlovid guideline have been presented in a basic form in a
Microsoft word document. Previously it was discussed that a Visio infographic was to be used,
however the Ministry Comms team believed a simple table in Word may be more useful. This
action is stil underway with the Comms team.
3.0
Therapeutics
1982
PHARMAC Update
• There are increased signs of usage of antiviral treatments:
ACT
o Increases have been seen since treatment criteria was widened (5 May 2022).
o Continued monitoring and data collection wil be used to determine if future updates to the
criteria are required.
o A web-based tool has been created to guide interpretation of the antiviral treatments
criteria. This is expected to be particularly useful in understanding how many risk factors
are required for access.
o The ‘He Ako Hiringa’ resource has been developed and released. This contains general
guidance on how to use and prescribe Molnupiravir and Paxlovid.
INFORMATION
• Evusheld (tixagevimab/cilgavimab) has been secured with likely availability from June 2022.
o Pharmac’s Treatment Advisory Group have discussed the eligibility criteria, and advice for
use is in progress.
o 20,000 courses have been secured with the option for an extension of a further 20,000
however, evidence is stil evolving on the duration of protection and timing of further doses
OFFICIAL
that may be required.
o Pharmac continues to seek advice from the COVID-19 Care in the Community team on
THE
serology to determine how this treatment wil work in a primary and secondary care
setting.
• Remdesivir stock for approximately 1,200 people is available, with ability to access more if
required.
• Baricitnib remains availabl
UNDER e, with stock available for approximately 330 people. It was noted there
has been very little use of this in the last few weeks.
• It is expected that more stock of tocilizumab wil be received.
o Ongoing work is in place with the supplier to understand when the stock wil arrive.
o An updated approach of managing ongoing supply is in progress.
o Assessment is required to determine how to restart treatments in patients who may have
had t
RELEASED o stop taking tocilizumab due to the supply issues.
Discussion:
• A member enquired whether there is any work correlating changes of prescribing and availability
of the oral antiviral treatments with patient outcomes or hospitalisation data.
o The COVID-19 Care in the Community team are working with the Data and Digital team to
provide this and are aiming for something to show by end of next week.
• It was raised that data had been presented for older people being prescribed Paxlovid and it was
asked whether Pharmac should consider lowering some of the thresholds for access, or if any
other fine tuning is required on the criteria for prescribing Paxlovid.
o Pharmac confirmed that they were continuing to collect data and feedback as the new
criteria is rolled out. This will be used to help them identify:
1. who it is getting treatment
2. who is missing out
3. a clearer picture of if any updates to the criteria are needed.
o It was noted that any updates to criteria wil be reviewed as identified.
• Discussion occurred on the use and importance of serology testing with regards to Evusheld.
It was noted:
o That advice has been given by the COVID-19 Testing TAG to formally go to the New
Zealand Microbiology Network for comment.
o Currently there is no clarity around how many people would be eligible for this medicine.
o That clarity is needed on the interpretation, practicality and logistics of serology testing as
1982
opposed to criteria that might rely on empirical views.
It was discussed that the serology could be used prophylactical y (e.g., transplant
patients that might be at high risk of severe outcomes from COVID
ACT -19 due to not
mounting an immune response to the vaccine might get Evusheld injections at the
appropriately determined frequency)
It was discussed that this would not be used for acute treatment under the current
Medsafe application.
o Data from the Auckland Hospital Renal Transplant study showed that the renal dialysis
and transplant cohort showed low serological conversion after second or third vaccines.
o Data at a national level would be helpful when determining the volume of patients that this
treatment could be appropriate for. (i.e., it could be clearly defined t
INFORMATION hat transplant patients
could benefit, but more information is required to determine which other specialties would
benefit, with mention of immunocompromised including haematological and rhetological
specialties.)
o A memo agreeing on risk factors would be of use.
o Providing information to doctors ahead of making Evusheld available would:
OFFICIAL
1. Allow time for patient identification
- Patient identification can be difficult in primary care and with large cohorts
THE
2. Allow serology testing to begin ahead of the approval
- This could help reduce the burden on serology labs when it is available
ACTION:
•
None stated. UNDER
Medsafe Update
• Actemra (tocilizumab) was approved on 12 May for use in COVID-19 hospitalised patients who are
receiving systemic corticosteroids and require supplemental oxygen or mechanical ventilations.
• Evusheld and Sotrovimab have both been approved in EU and Australia.
• A meeting w
RELEASED as held with Gilead to discuss Remdesivir. They are expected to respond to a request
for further information by end of June. s 9(2)(b)(ii)
STA Update
• Highlights from the latest developments in science were presented. This included:
o A deep dive on Paxlovid trials in vaccinated patients.
It was estimated that vaccination reduces risk of hospitalisation by 35%, and new
numbers needed to treat (NNT) have been produced
Based on the adjusted risk:
• EPIC-HR trial (Paxlovid) NNT will increase from 18 to 27
• MOVe-OUT trial (Molnupiravir) NNT will increase from 34 to 53
o A deep dive assessed pre-exposure prophylaxis and post-exposure prophylaxis of
Paxlovid
The trial did not meet its primary end point (i.e., it did not significantly reduce
household infections when used as a post-exposure prophylaxis)
o There were no changes in the international approved treatments lists.
• An update on the data for risk of hospitalisation data for the heat map and access criteria was
given:
o The Ministry’s Insights team have worked in this space and the final data is in progress
towards sign-out. Data should be ready for the Therapeutics TAG by end of next week for
crude and adjusted risk ratios for standard demographic factors and vaccination for
population level risk and case level risk.
Discussion:
1982
• It was noted that this data should connect with data from the COVID-19 Care in the Community
work.
ACT
ACTION:
- STA data team and COVID-19 Care in the Community to liaise and align col ected data.
Primary Care Update
o An update accompanied by slides was presented: Data showing numbers of Molnupiravir
and Paxlovid prescribed to date:
The access criteria widened on the 5 May. Increased rates of prescribing were
INFORMATION
seen within 24 hours of the criteria widening. This has been maintained since, with
dips in prescriptions over the weekends.
The rates of Molnupiravir and Paxlovid prescribed appear to track in parallel with
each other (i.e., there has not yet been an increased proportion of Molnupiravir to
Paxlovid seen).
OFFICIAL
o Prescription data was broken down according to ethnicities:
Work is being completed with the Data and Digital team as this could be used to
see if the rates pr
THE escribed match the prevalence of COVID-19 in the community
(i.e., used as a measure of equity).
Rate in prescribing to Māori appear to be increasing
No increases seen in Pacific peoples.
o Data on the dispe
UNDER nsing rates of Paxlovid for its first month (4 April to 5 May):
Prescribing rates for older Māori and Pacific peoples were higher than
Asian/European.
This was expected given the increased risks in these populations
A deeper dive is to be completed as more information is collected.
o Data on prescriptions was broken down by deprivation status:
Deprivation appears to be a strong indicator of hospitalisation risk
RELEASED The data showed a peak in the most deprived being prescribed the most recently.
It was noted this is encouraging, however not a sustained pattern yet.
The least deprived group were often among those most prescribed to.
It was noted that the deprivation data should be interpreted with caution as it can
be misleading due to a lot of factors contributing differences within deprivation
status. One point mentioned was that wealthier people tended to live longer and
older people were identified more as requiring therapeutics. This could be a
contributor to why we are see high prescribing in higher deprivation groups.
o Data of prescriptions per DHB:
It was noted this should be led by where the outbreak is more dominant
Noted that the data of DHB with highest rates of infection were not aligned with
where the highest prescribing was seen.
It was discussed that high rates of infection are not always an indicative of
hospitalisation and severe disease, therefore it was of limited data using rates of
infection as a main metric for comparing rates of prescriptions too.
o Data from an RNZCGP survey on barriers to prescribing was presented:
This identified complicated access criteria (proactively promoting Pharmac’s
criteria tools to assist with prescribing)
Approximately 50% of eligible patients were perhaps unnecessarily excluded due
to contraindication and drug interactions
Other identified barriers included: insufficient time/resources/lack of systemic
support identifying the vulnerable/eligible.
Key ways to support barriers to prescribing were identified:
1982
•
Encouraging pharmacists and specialists to work closely with GPs to
support their decision making
•
Primary Health Organisations (PHOs) are helping to suppor
ACT t
•
The National Risk Stratification tool is not yet accurate enough to support
the decision making of who can be proactively identified.
o The Care in the Community team are looking at equitable outcomes using data,
communication plans (through multi-pronged approach to people directly and the health
sector) and raising awareness (through webinars).
Risk scores can be another facilitator aiding people to get access to therapeutics
Meetings with Global Health, Pharmac and MFAT to understand how to donate
INFORMATION
therapeutics to the Pacific are underway.
o Other:
The team is continuing to monitor utilisation against supply (with Pharmac
support), actively encouraging review of the eligibility criteria/access barriers
Working with Pinnacle PHO to conduct an audit of:
•
Who met access criteria
OFFICIAL
•
Who tested positive
•
How many people received therapeutics
THE
He Ako Hiringa published a practice-level self-audit tool for Paxlovid prescribing.
They are also developing a communications plan, which involves liaising with Iwi and
Pacific community providers, Equity and Treaty responsiveness team, Disability support
providers, Care coordination hubs and Interagency partners, to ensure the work is
UNDER
targeted to priority and vulnerable populations.
Discussion:
•
Information on the number of prescriptions divided by DHB needs more information to get a full
picture. Suggestions included:
o Dataset to account for size of DHB
o Datas
RELEASED et to account for demographics of DHB
o Data to capture DHB end goals. (i.e., the DHBs with highest infection rates also had very
low or no hospitalisation/deaths)
More work is being done by Data and Digital to add strength to this dataset
It seems likely that some DHBs are likely still under-utilising therapeutics
•
STA noted that MoH have a Behaviour Science team conducting surveys within the population
with similar questions as asked in the RNZCGP survey.
o It was noted that now therapeutics are available it would be good to get questions about
them addressed to the general public as well as health providers.
• It was clarified that the Health Pathway Webinar is to talk to:
o The 50% contraindication to spread key messaging, with follow up survey to see if this has
an impact on educating.
o Drug interaction and working with pharmacists, (i.e., spread the work, col aboration key)
o Treaty advisor to raise awareness of barrier to access.
o It was noted that many ED practitioners firmly believe antiviral treatments in scope of GP.
This would be an area to explore to increase prescribing as:
Could be an issue as many people use ED in lieu of GP services
ED staff are able to prescribe (particularly promoted in rural areas)
Noted that there is a risk due additional monitoring required on antiviral treatment
and good process are required and in place
After diagnosis in ED the case needs to be linked into the GP and ED Eclair
notification can take 3 days so this may take them out of time.
ACTION
: Memo to specialist for Antiviral (Elaine to work on)
1982
• Data team with STA to link in with COVID in the Community team to have fullest utilisation as
possible
ACT
4.0
Equity Considerations
• ED Eclair notification can take three days and therefore patients presenting at later stages can
miss the window of opportunity for treatment. This could include high-risk people who would
benefit from treatment.
• Good to see meaningful response from GPs on barriers to prescribing
• Support in prescribing was discussed:
INFORMATION
o Support differs between clinics
o Update with Health Pathways link to Pharmac has been helpful for prescribers.
Discussion:
None noted
OFFICIAL
5.0
Antiviral Access Criteria Update
None noted
THE
6.0
Future of Therapeutics TAG
Scope and context of reforms
UNDER
• The Group were thanked for their contributions to date. With the STA’s move to the Public Health
Agency, there needs to be a conversation about where is the best place for the Therapeutics TAG
to sit.
• The original mandate of the Group was to develop and support hospital guidelines and to provide
advice to ministry.
• The Group’s role has expanded to provide advice and support of community guidelines, whether
RELEASED
treatments are reaching the right people and whether there are gaps
Core elements to keep for success for the future:
• Need to keep the Therapeutic TAG sustainable for all involved, therefore there was a suggestion
to move to monthly meetings, with an ability to meet more rapidly if required for a particular reason
• Suggested to discuss further at the next Therapeutic TAG meeting, as several member were
apologies for the current meeting.
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
3.0
Therapeutics
PHARMAC Update
• Evusheld: Pharmac have secured a supply of Evusheld (AstraZeneca’s monoclonal antibody).
This is expected to be available later this month or early July, dependant on Medsafe approval.
o Pharmac have been seeking advice from their COVID-19 technical advisory group and
external experts on the possibility of using serology within the access criteria. Based on
this advice, this is not being pursued initially due to various complications associated.
Alternatively, a list wil be provided of the various conditions or situations that could
mean someone is severely immunocompromised, as has been used by other
jurisdictions overseas.
o The access criteria are still subject to a consultation, which is expected to be released
next week. Feedback wil be welcomed on criteria and whether the conditions and
groups targeted are captured correctly.
o Pharmac are working closely with CinC to determine how the implementation of
1982
Evusheld wil work across primary and secondary care settings.
• Oral antiviral treatments (Paxlovid and Molnupiravir): Pharmac are working closely with CinC to
ACT
monitor usage and to update the criteria as new data, evidence and usage trends emerge.
• Remdesivir: There is stock for approximately 1100 people available, and more stock can be
obtained from the supplier as needed. Advice has been received from the Pharmac COVID-19
advisory group that there is interest in widening the criteria. This is something that may be
progressed after Evusheld is completed.
• Baricitinib has had a low uptake in usage over the last few weeks. Currently there is stock for
approximately 330 people stil available.
• Tocilizumab: stock has started to be received however the stock level requires close
INFORMATION
management as the pandemic is continuing.
Discussion:
Evusheld
• Members noted that it would be useful to see some data about severely immunocompromised
OFFICIAL
people. However, is appreciated that the people who are best placed to col ate this data would
find this difficult in the current clinical setting.
THE
• It was discussed that it would be helpful for providers to receive more information from CinC
relating to the implementation, in particular the expectations about delivery and supply of
Evusheld.
o Currently there is no plan signed off on the implementation process, however the
proposal is for a
UNDER joint effort between primary, community, and secondary care.
o This would likely have an emphasis on secondary care capturing a large number of the
urban and community population to ensure that more rural or remote people who are
not regularly linked into outpatient clinics also get access.
o Secondary care has the advantage of having full clinics of people that meet the eligibility
criteria.
• Pharmac are currently not drafting serology into the criteria but welcome feedback if a use is
RELEASED
identified. It was noted that the criteria may widen to more groups as more stock becomes
available.
Medsafe Update
• The initial evaluation for Evusheld has been completed and Medsafe are expecting the
additional requested information from the company by 17 June.
• The initial evaluation for Sotrovimab has been partial y completed. A request for further
information has been requested on quality control.
STA Update
• Two pre-prints about the real-world evidence of effectiveness of Molnupiravir and Paxlovid have
been published. These have reported that:
o both Molnupiravir and Paxlovid reduced disease progression in moderately il in-patients
who were not on supplemental oxygen
o Paxlovid (but not Molnupiravir) reduced the risk of hospitalisation in out-patients.
• Evusheld (AZD7442) retains neutralising ability against BA.4 and BA.5.
• Rebound infections have been associated with Paxlovid. Reports of rebound infectious are
increasing, but as yet none have reported severe disease. The CDC recommends anyone
experiencing a rebound in COVID-19 symptoms after the completion of a 5-day Paxlovid course
should isolate for a further 5 days (but does not recommend a second course). COVID-19
monoclonal antibodies have significantly longer half-lives and bioavailability than antivirals,
1982
hence are less susceptible to rebound.
• RCT (ACTT-4) reports no significant difference in mortality, clinical progression, use of invasive
(and non-invasive) ventilation between dexamethasone and baricitinib in hospital
ACT ised
individuals; baricitinib had a better safety profile.
Discussion:
• It was noted that although there may be some equivalence between dexamethasone and
baricitinib, there is a significant difference in their costs.
o Some people experience significant harm from treatment with dexamethasone,
particularly people with diabetes.
o Careful considerations between risks and benefits are needed bef
INFORMATION ore prescribing
dexamethasone.
o It was also acknowledged that dexamethasone is significantly more affordable.
Primary Care Update
• Data was presented on prescribing of oral therapeutics, showing that rates in prescriptions are
OFFICIAL
rising as prescribers become more familiar with the therapeutics. It was noted that the number of
prescriptions understandably dips during the weekends.
THE
• CinC are exploring ways to overcome prescribing barriers that represent workforce availability.
Additional planning is underway for winter and bigger surges, and how the stresses from this to
the healthcare system can be managed.
• Pharmacist prescribers can now prescribe oral therapeutics.
• Overall, Europeans are c
UNDER urrently receiving the largest numbers of prescriptions. Pacific peoples
have had the highest numbers of Paxlovid prescriptions per proportion of active cases, with
1.5% of Pacific peoples who become a case receiving a course of Paxlovid within 5 days. Māori
have been receiving Molnupiravir at a much higher rate than other ethnicities in last few days.
• As expected, older people are receiving drugs at greater rate than younger. There has been
higher prescribing for older Māori than Pacific peoples or Europeans. Asian currently have the
highest rat
RELEASED es of prescription in the 85-89 age bracket in the past week. These trends wil need to
be monitored to help detect divergence between groups over time.
• In terms of deprivation status, people in the least deprived quintile have been receiving a greater
portion of oral therapeutics than the other deprivation groups. It is unclear at this stage if this
bias is due to people being less deprived being more likely to live to an older age. Al other
deprivation statuses appear to be prescribed to in an equal amount.
• Data has been col ected on the number of prescriptions in each DHB. Currently Canterbury is
ahead which is reflective of the high rates of infection in the region. Auckland, Southern and
Waikato all also had high rates of prescription.
• CinC are conducting regular meetings with equity advisors to ensure hard to reach populations
have awareness and access to these drugs. Communications are being produced in different
languages and in accessible formats to ensure as many people can access them as possible.
Additionally, resources and webpages are being developed specifical y for the providers of
disability services to help with funding applications and to improve accessibility for people in
their care.
Discussion:
• It was noted that primary care colleagues could find it useful to see information and specific data
from countries already successful y using the therapeutics.
o CinC indicated that they are intending to do this.
4.0
Equity Considerations
• No new information was discussed. It was noted that there had been a lot of mahi in putting
equity at the core of the CinC work.
1982
5.0
Future of Therapeutics TAG
ACT
• There is an ongoing need to discuss how to make sure the group remains sustainable moving
forwards, what purpose the group serves and for whom, the scope of requests that the TAG is
used for, and how advice should be commissioned through the group.
• Monthly meetings were discussed as an ideal option, with some flexibility to deal with bigger
issues when they arise.
• It was noted the appropriate levels of support required by the group moving forwards would
need to be discussed and determined.
INFORMATION
Monkeypox
• The response to Monkeypox has shown a potential strength and future utility of the group. It was
possible to turn around hugely valuable advice in a quick timeframe partly because the group is
already established and ready to go.
Discussion:
OFFICIAL
• The possibility of the Therapeutics TAG moving away from specific responses and setting up an
emerging infectious diseases res
THE ponse was discussed.
o This included suggestion of a high consequences infectious disease system where all
emerging diseases have same people set up and ready to respond.
o An example of this has been seen in the UK.
o It was noted that some flexibility to the scope/agenda would al ow the TAG to deal with
UNDER
bigger topics when they arose would be helpful.
o These wil be part of ongoing discussions as the new entities come into place, with
feedback sought on the advice needed from within the Ministry.
6.0
Hospitalisation risk data
Preliminary data from the Ministry’s Science, Surveil ance and Insights team was presented that looks at
RELEASED
an epidemiological analysis of the risk of a COVID-19 cases becoming hospitalised and the associated
mortality risk estimates.
It was noted that there is now a new larger hospitalisation dataset (the National Minimum) which was
used.
• The data includes hospitalisations from a list of causes that might be related to COVID-19.
• It then excludes anyone who was known to be not a COVID-19 case.
•
The definitions are very wide, so it is expected that approximately 75% of the dataset are
possibly COVID-19 related.
•
Further updates to the dataset are expected.
The presentation showed:
•
The cumulative incidences for hospitalisation amongst cases.
•
The incidence ratios between groups including adjusted risk.
•
Data broken down by basic demographics, including gender, ethnicity, deprivation status and
vaccination status. It was noted that:
o at this stage the vaccination status had not been adjusted to account for the timing of
the vaccination, although this is something that wil be completed
o Pacific peoples appear to have higher risk than other ethnicities even when deprivation
status is considered
•
The next steps are to include some stratification to see what is happening within the adjusted
risk models. This wil be completed by adding morbidity measures, such as M3 and P3 scores
1982
while considering timing since vaccination.
Discussion:
ACT
•
The dataset was considered very valuable for discussions of who is at risk, which has been an
ongoing concern of the Therapeutics TAG.
•
It was noted that the total numbers of COVID-19 deaths reported do not appear to match the
numbers of COVID-19 deaths at hospital. It was speculated this is likely due to elderly dying at
home or in rest homes.
o Although the dataset captures mortality rates and hospitalisation dates, it does not
record where a case has died.
INFORMATION
•
There is potential to import a model of therapeutics use into the dataset.
It may be possible to run a model comparing people who did get therapeutics
with people who did not to see if this changed the risk of hospitalisation. It was
noted that it could be chal enging to get perfectly matched groups of people who
did and did not get therapeutics due to the complex eligibility criteria.
o Additionally, hospitalisation and any subs
OFFICIAL equent length of hospitalisation as wel as
mortality can be investigated between these two groups.
o Therapeutics models would likely rely on data relating to dispensed therapeutics versus
THE
an outcome.
It was cautioned that non-adherence would not be considered in these models
and would be a limitation.
This is a common limitation to epidemiology studies, however, this data would
UNDER
provide an indication when used together with other evidence.
o It was noted that it would be of interest to check what therapeutic data-points can be
accessed.
The data wil be brought back to the Therapeutics TAG once this further information has been collated
and analysed.
RELEASED
7.0
Guideline Updates
•
No updates for this meeting, however, it was noted that the next planned update to the
Guideline was scheduled for the end of this month. Evusheld is the next item which will require
updates to allow it to be included in the in-hospital guidance, and the group are not expecting
any other major changes.
Memo on antivirals
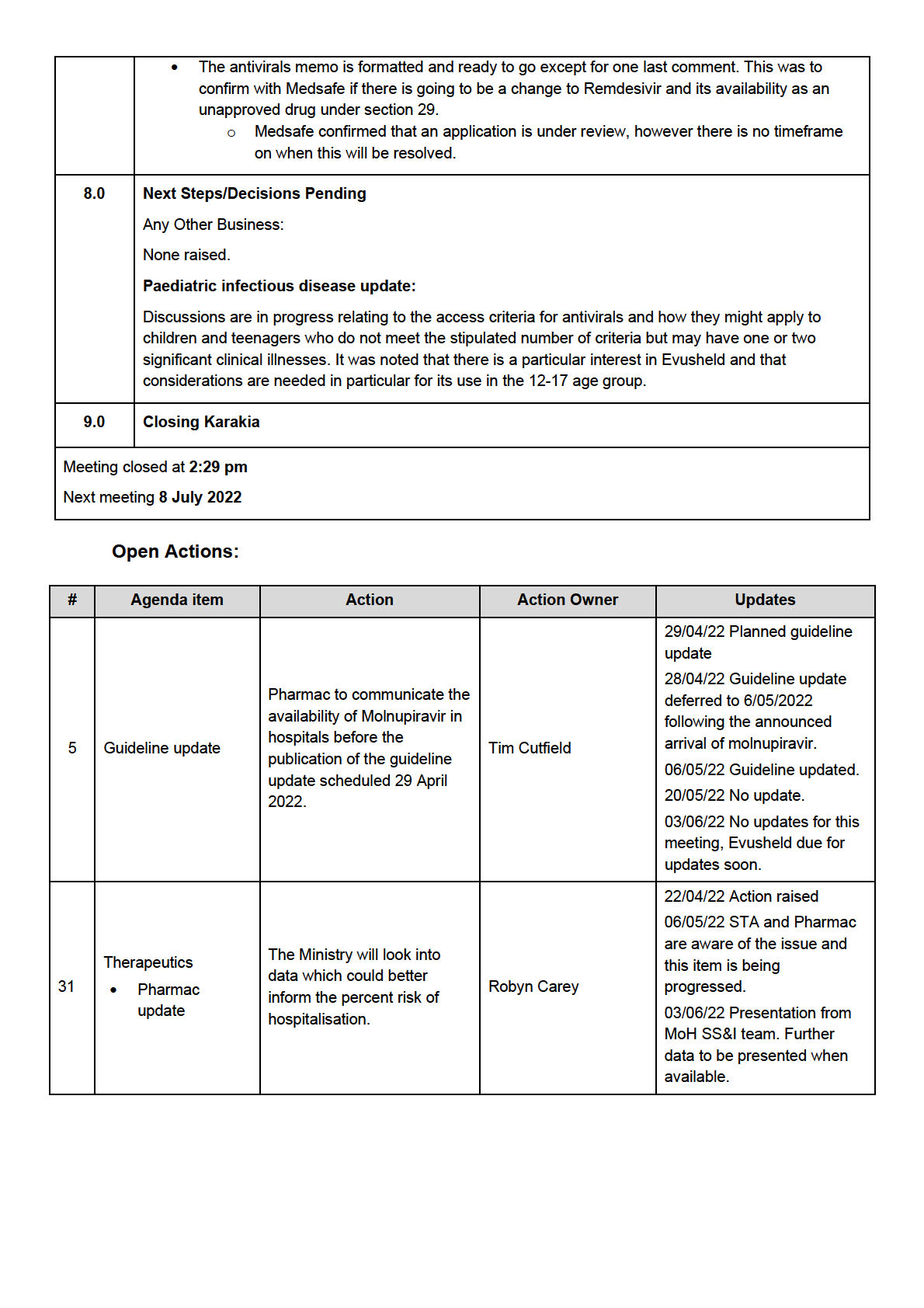 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
• 216 vials of Evusheld are available for use now and can be ordered from Healthcare Logistics
(HCL). These have no current access criteria, however Pharmac have made suggested criteria
available.
• There is ongoing review of antiviral access criteria being completed in collaboration with COVID
care in the community (CinC) team.
• Remdesivir has been added to the Pharmac schedule under section H (hospital use).
• Tocilizumab stock levels and management is returning to normal and the patients who were
taken off this treatment due to supply issues may be changed back to it if required.
• Baricitinib stil has enough stock for approximately 330 patients.
A member queried how the remdesivir stock supply is going and whether there was enough stock for
new surges. Pharmac noted that there is currently about 800 patient’s worth of stock available. Pharmac
can access more stock if required and are likely to look into doing this.
Medsafe Update
Medsafe evaluations are nearing completion for both Evusheld and sotrovimab. It is expected that
1982
decisions on whether consent can be given for both drugs and if either wil need to be referred to
Medicines Assessment Advisory Committee are likely to be made within the next week. If these do go to
ACT
the committee, this wil be meeting later this month.
STA Update
• An update was given by the STA representative. This matched the information circulated with
the agenda.
Primary Care Update
• It was noted that pharmacists wil soon be able to prescribe Paxlovid and molnupiravir.
INFORMATION
• Due to changes in staff, specific questions relating to primary care wil be required ahead of
meetings to help staff prepare.
4.0
Equity Considerations
An update was requested in relation to the improvements of access of oral antiviral in the community,
OFFICIAL
specifical y relating pharmacists being able to prescribe oral vials and improve weekend access.
• It was reported that work is in progress, with advice being sought from the successful
THE
implementation of prescriptions through community pharmacies in Quebec.
• A mechanism is being put in place to reclassify medicines temporarily and rapidly to pharmacist
use.
UNDER
5.0
Update on Hospitalisation and Mortality Data
Data on the risks of hospitalisation and mortality associated with COVID was presented the TAG by a
guest from the Intelligence, Surveil ance and Knowledge team. This built on the presentation shown at
the previous TAG.
• Data relating to hospitalisation was restricted to cases in which COVID-19 could not be ruled out
as a cause/
RELEASED contributor.
• Data relating to mortality included cases which can be attributed to COVID-19 as an underlying
or contributing factor to death.
• Mortality report uses different age split to usual COVID-19 data sets due to the increased
association between age and mortality.
Overall, in terms of mortality:
• There had been relatively low rates in people over 60 prior to March 2022
• Generally decreasing rates for most age groups since the first wave. However, this is not true for
the older age groups:
o For 80-89 and 90+ year olds mortality has continued to rise
• In the latest data, significant increases in 65+ age group
o Approximately 50% increase in Maori
There is the potential to investigate the link between hospitalisation and the potential for therapeutic
intervention.
A preliminary look at data has investigated the window of time from onset of symptoms or COVID-19
report date to hospitalisation
• Minimal numbers of people reported symptoms prior to hospitalisation with most people self-
reporting symptom onset as the day of hospitalisation.
• Data surrounding the onset of symptoms was only recorded for 50% of hospitalisation cases.
From this:
o Approximately 25% had symptoms start on the day of hospitalisation (Day 0)
1982
o Approximately 50% reported symptoms within days 1-5
o Approximately 20% reported symptoms after more than 5 days.
ACT
• Risk factors for hospitalisation showed vaccination was the strongest modifiable risk factor for
people going to hospital. Increased age and co-morbidities were also strong indicators of
hospital risk.
Action: To share slides with the Therapeutics TAG.
6.0
Query about change in access criteria to primary care
Criteria for Paxlovid use based on Canadian experience have been included in the hospital guideline to
INFORMATION
allow consideration of an option for its use in people with severe renal impairment (eGFR<30 or on
dialysis) in a hospital setting. This has led to a discussion on if this widening is also appropriate to
extend to a primary care setting and if so, what are the appropriate safeguards required.
Rational for the extension to a hospital setting was:
• The initial exclusion of people with advanced kidn
OFFICIAL ey disease from treatment with Paxlovid was
that there was no evidence relating to this group from initial Paxlovid validation trails.
o This is likely due to this group presenting as high risk during the initial phase of clinical
THE
trial where safety and efficacy are stil being proved.
• Chronic kidney disease is frequently a marker of multi-morbidity and high risk from COVID-19
• Other countries have working groups that have put together a proposal for dosing based on
small numbers of a pharmacokinetics study that suggest dosing is appropriate for people with
UNDER
advanced renal dysfunction, including dialysis.
• This is a ‘consider use’ as there is not yet data from a large-scale study supporting the safely,
however there is a compel ing argument that the from early phase 1 trials that this is safe and
tolerable.
• Currently the only other COVID-19 antiviral treatments available to these patients in NZ are
remdesivir and molnupiravir. However, with remdesivir the excipient betadex sulfobutyl ether
RELEASED
sodium is renally cleared and accumulates in patients with decreased renal function; requiring
caution if eGFR is less than 30 mL/min/1.73m2 (limited information available). Molnupiravir can
be used, but from published studies is expected to be considerably less effective. Therefore, the
NZ guideline group felt that it was reasonable to fol ow other countries and recommend
“consider use” of Paxlovid for patients in these groups that are in hospital.
• Use in the hospital may be the appropriate place to build experience with this treatment initially.
• This could be a way to treat people who may otherwise be excluded from efficacious early
treatment options, however some safety concerns are appropriate.
•
Noted the dispensing issues (day night, half doses)
The regulator point of view is that as this is prescribed under Section 29, the medical practitioner would
have to make a professional judgement that its use is appropriate.
The TAG noted the fol owing in relation to providing advice for use of Paxlovid in primary care:
•
It may be reasonable and beneficial for people with eGRF<30 to be considered for treatment
with Paxlovid.
•
Providing clear guidance would be more useful than a generic ‘discuss with
GP/nephrologist/infectious disease physician’. This is particularly relevant in smal er centres
where no/less specialists are on hand.
•
The logistics of dispensing the drug at an appropriate and easy to use way for the patient needs
to be considered.
•
Support required to pharmacist on the chal enges of the treatment and adjustments to dose.
o May result in some parts of packs being disposed of due to adjusted dose
•
In summary, it seems reasonable to provide in a primary care setting and may prove to be
1982 less
of a safety issue and more of a dispensing problem.
Action: To draft guidelines for GPs and seek Medsafe advice.
ACT
7.0
Fully vaccinated definition and access criteria
It has been noticed that some hyperlinks in guidelines currently lead to pages that no longer exist. In
particular, the page that defined ‘fully vaccinated’ has been removed. The concept of ‘fully vaccinated’ is
changing as new evidence emerges throughout the pandemic.
The discussion relates to what is the threshold with regards to vaccination where risk shifts from a lower
risk to higher and where the threshold lies in access to therapeutics.
INFORMATION
It was noted that the COVID-19 Vaccine TAG (CV TAG) is working on a definition of fully vaccinated
Action: To check in with CV TAG for their definition and ensure this is relevant in therapeutics.
8.0
Guideline Updates
OFFICIAL
•
The updates to hospital guideline include:
o The use of Paxlovid in hospital setting (discussed in item 6.0).
THE
o A definition of ‘fully vaccinated’ (discussed in item 7.0).
o Support for using remdesivir for people with moderate COVID-19 who are early in il ness
(based on the Solidarity trail results)
o Evusheld referenced in guideline.
o A recommendat
UNDER ion not to use baricitnib instead of steroids (based on current limited
supply, cost, availability). This could be reassessed if changes to stock are implemented
by Pharmac.
Action: To draft a memo with support from the Therapeutics TAG.
9.0
New structure of MOH/PHA
RELEASED
A presentation was given to explain the new structure of the Health Teams:
•
Health NZ: providing the operational and delivery roles. (This includes the National Public Health
Service Organisation)
•
Ministry of Health: providing the chief stewardship role (i.e., performance, regulation, advice to
the minister etc.)
In total there wil be nine directorates that make up the Ministry of Health (These are: Public Health
Agency (PHA), Evidence Research and Innovation (ERI), Strategy and Policy, Maori Health and Maori
Crown relations, Regulations, System Performance and Monitoring, Government and Executive
Services, Corporate Services and Clinical leadership)
Key take home:
• Science Technical and Advisory (STA) team will stay at the MoH within PHA directory
• PHA includes: the Intelligence, Surveil ance and Knowledge team (this is made up of STA,
Intel igence and analytics and some broader communicable disease teams).
• The ERI may also be of interest to TAG members. This is a completely new directorate which
will working with STA on many pieces of advice.
10.0
Next Steps/Decisions Pending
Monkeypox update:
• There has of yet been no probable or confirmed case of Monkeypox in New Zealand, as of the
current date (8th July).
1982
• A test definition has been agreed on and monkeypox has been made a notifiable disease.
• The advice provided by this TAG was to use vaccination as a strategic approach, targeting close
contacts post-exposure rather than a broader public health vaccination.
ACT
• New Zealand government is looking at options to secure the third-generation vaccine. Of note
the current stock is second-generation and expired
• There is a smal pool of stock of cidofovir. Trying to get access to tecovirimat, however this is
limited internationally.
• Work is underway to get access to vaccinia immune globulin and antivirals from Australia if
required. New Zealand is in the queue for next year, although exact quantities has been
requested.
INFORMATION
• A request for advice has been received around pre-exposure prophylaxis for specific groups
(i.e., MSM) and if this is relevant in a New Zealand context. Discussion with CV TAG members
suggested use post exposure for close contacts may be more appropriate (pending international
experience).
11.0
Closing Karakia
OFFICIAL
Meeting closed at
2:53 pm
THE
Next meeting
05 August 2022
UNDER
RELEASED
 Open Actions:
Open Actions:
#
Agenda item
Action
Action Owner
Updates
Paxlovid use in primary To draft guidelines for GPs
33
Jennifer Keys/STA
817122 Item opened
care
and seek Medsafe advice.
To check in with CV TAG for
Fully vaccinated
their definition and ensure
34
definition and access
STA
817122 Item opened
this is relevant in
criteria
Therapeutics.
To draft a memo with
35
Evusheld advice
support from the
ST A/Tim Cutfield
817122 Item opened
Therapeutics TAG.
1982
Closed Actions:
ACT
#
Agenda item
Action
Action Owner
Updates
29/04/22 Planned guideline update
28/04/22 Guideline update deferred
to 6/05/2022 following the announced
Pharmac to communicate the
arrival of molnupiravir.
availability of Molnupiravir in
06/05/22 Guideline updated.
INFORMATION
hospitals before the
5
Guideline update
Tim Cutfield
20/05/22 No update.
publication of the guideline
03/06/22 No updates for this
update scheduled 29 April
meeting, Evusheld due for updates
2022.
soon.
08/07/2022 Update to guidelines was
OFFICIAL
given. Action closed as now standing
item.
THE
22/04/22 Action raised
06/05/22 ST A and Pharmac are
aware of the issue and this item is
being progressed.
UNDER
The Ministry will look into
Therapeutics
data which could better
03/06/22 Presentation from MoH
31
Robyn Carey
inform the percent risk of
SS&I team. Further data to be
Pharmac update
presented when available.
hospitalisation.
08/07/2022 Updated presentation on
hospitalisation and mortality data was
given. To share slides with
RELEASED
Therapeutics TAG. Item closed.
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
• 216 vials of Evusheld are available for use now and can be ordered from Healthcare Logistics
(HCL). These have no current access criteria, however Pharmac have made suggested criteria
available.
• There is ongoing review of antiviral access criteria being completed in collaboration with COVID
care in the community (CinC) team.
• Remdesivir has been added to the Pharmac schedule under section H (hospital use).
• Tocilizumab stock levels and management is returning to normal and the patients who were
taken off this treatment due to supply issues may be changed back to it if required.
• Baricitinib stil has enough stock for approximately 330 patients.
A member queried how the remdesivir stock supply is going and whether there was enough stock for
new surges. Pharmac noted that there is currently about 800 patient’s worth of stock available. Pharmac
can access more stock if required and are likely to look into doing this.
Medsafe Update
Medsafe evaluations are nearing completion for both Evusheld and sotrovimab. It is expected that
1982
decisions on whether consent can be given for both drugs and if either wil need to be referred to
Medicines Assessment Advisory Committee are likely to be made within the next week. If these do go to
ACT
the committee, this wil be meeting later this month.
STA Update
• An update was given by the STA representative. This matched the information circulated with
the agenda.
Primary Care Update
• It was noted that pharmacists wil soon be able to prescribe Paxlovid and molnupiravir.
INFORMATION
• Due to changes in staff, specific questions relating to primary care wil be required ahead of
meetings to help staff prepare.
4.0
Equity Considerations
An update was requested in relation to the improvements of access of oral antiviral in the community,
OFFICIAL
specifical y relating pharmacists being able to prescribe oral vials and improve weekend access.
• It was reported that work is in progress, with advice being sought from the successful
THE
implementation of prescriptions through community pharmacies in Quebec.
• A mechanism is being put in place to reclassify medicines temporarily and rapidly to pharmacist
use.
UNDER
5.0
Update on Hospitalisation and Mortality Data
Data on the risks of hospitalisation and mortality associated with COVID was presented the TAG by a
guest from the Intelligence, Surveil ance and Knowledge team. This built on the presentation shown at
the previous TAG.
• Data relating to hospitalisation was restricted to cases in which COVID-19 could not be ruled out
as a cause/
RELEASED contributor.
• Data relating to mortality included cases which can be attributed to COVID-19 as an underlying
or contributing factor to death.
• Mortality report uses different age split to usual COVID-19 data sets due to the increased
association between age and mortality.
Overall, in terms of mortality:
• There had been relatively low rates in people over 60 prior to March 2022
• Generally decreasing rates for most age groups since the first wave. However, this is not true for
the older age groups:
o For 80-89 and 90+ year olds mortality has continued to rise
• In the latest data, significant increases in 65+ age group
o Approximately 50% increase in Maori
There is the potential to investigate the link between hospitalisation and the potential for therapeutic
intervention.
A preliminary look at data has investigated the window of time from onset of symptoms or COVID-19
report date to hospitalisation
• Minimal numbers of people reported symptoms prior to hospitalisation with most people self-
reporting symptom onset as the day of hospitalisation.
• Data surrounding the onset of symptoms was only recorded for 50% of hospitalisation cases.
From this:
o Approximately 25% had symptoms start on the day of hospitalisation (Day 0)
1982
o Approximately 50% reported symptoms within days 1-5
o Approximately 20% reported symptoms after more than 5 days.
ACT
• Risk factors for hospitalisation showed vaccination was the strongest modifiable risk factor for
people going to hospital. Increased age and co-morbidities were also strong indicators of
hospital risk.
Action: To share slides with the Therapeutics TAG.
6.0
Query about change in access criteria to primary care
Criteria for Paxlovid use based on Canadian experience have been included in the hospital guideline to
INFORMATION
allow consideration of an option for its use in people with severe renal impairment (eGFR<30 or on
dialysis) in a hospital setting. This has led to a discussion on if this widening is also appropriate to
extend to a primary care setting and if so, what are the appropriate safeguards required.
Rational for the extension to a hospital setting was:
• The initial exclusion of people with advanced kidn
OFFICIAL ey disease from treatment with Paxlovid was
that there was no evidence relating to this group from initial Paxlovid validation trails.
o This is likely due to this group presenting as high risk during the initial phase of clinical
THE
trial where safety and efficacy are stil being proved.
• Chronic kidney disease is frequently a marker of multi-morbidity and high risk from COVID-19
• Other countries have working groups that have put together a proposal for dosing based on
small numbers of a pharmacokinetics study that suggest dosing is appropriate for people with
UNDER
advanced renal dysfunction, including dialysis.
• This is a ‘consider use’ as there is not yet data from a large-scale study supporting the safely,
however there is a compel ing argument that the from early phase 1 trials that this is safe and
tolerable.
• Currently the only other COVID-19 antiviral treatments available to these patients in NZ are
remdesivir and molnupiravir. However, with remdesivir the excipient betadex sulfobutyl ether
RELEASED
sodium is renally cleared and accumulates in patients with decreased renal function; requiring
caution if eGFR is less than 30 mL/min/1.73m2 (limited information available). Molnupiravir can
be used, but from published studies is expected to be considerably less effective. Therefore, the
NZ guideline group felt that it was reasonable to fol ow other countries and recommend
“consider use” of Paxlovid for patients in these groups that are in hospital.
• Use in the hospital may be the appropriate place to build experience with this treatment initially.
• This could be a way to treat people who may otherwise be excluded from efficacious early
treatment options, however some safety concerns are appropriate.
• Noted the dispensing issues (day night, half doses)
The regulator point of view is that as this is prescribed under Section 29, the medical practitioner would
have to make a professional judgement that its use is appropriate.
The TAG noted the fol owing in relation to providing advice for use of Paxlovid in primary care:
• It may be reasonable and beneficial for people with eGRF<30 to be considered for treatment
with Paxlovid.
• Providing clear guidance would be more useful than a generic ‘discuss with
GP/nephrologist/infectious disease physician’. This is particularly relevant in smal er centres
where no/less specialists are on hand.
• The logistics of dispensing the drug at an appropriate and easy to use way for the patient needs
to be considered.
• Support required to pharmacist on the chal enges of the treatment and adjustments to dose.
o May result in some parts of packs being disposed of due to adjusted dose
• In summary, it seems reasonable to provide in a primary care setting and may prove to be
1982 less
of a safety issue and more of a dispensing problem.
Action: To draft guidelines for GPs and seek Medsafe advice.
ACT
7.0
Fully vaccinated definition and access criteria
It has been noticed that some hyperlinks in guidelines currently lead to pages that no longer exist. In
particular, the page that defined ‘fully vaccinated’ has been removed. The concept of ‘fully vaccinated’ is
changing as new evidence emerges throughout the pandemic.
The discussion relates to what is the threshold with regards to vaccination where risk shifts from a lower
risk to higher and where the threshold lies in access to therapeutics.
INFORMATION
It was noted that the COVID-19 Vaccine TAG (CV TAG) is working on a definition of fully vaccinated
Action: To check in with CV TAG for their definition and ensure this is relevant in therapeutics.
8.0
Guideline Updates
OFFICIAL
• The updates to hospital guideline include:
o The use of Paxlovid in hospital setting (discussed in item 6.0).
THE
o A definition of ‘fully vaccinated’ (discussed in item 7.0).
o Support for using remdesivir for people with moderate COVID-19 who are early in il ness
(based on the Solidarity trail results)
o Evusheld referenced in guideline.
o A recommendat
UNDER ion not to use baricitnib instead of steroids (based on current limited
supply, cost, availability). This could be reassessed if changes to stock are implemented
by Pharmac.
Action: To draft a memo with support from the Therapeutics TAG.
9.0
New structure of MOH/PHA
RELEASED
A presentation was given to explain the new structure of the Health Teams:
• Health NZ: providing the operational and delivery roles. (This includes the National Public Health
Service Organisation)
• Ministry of Health: providing the chief stewardship role (i.e., performance, regulation, advice to
the minister etc.)
In total there wil be nine directorates that make up the Ministry of Health (These are: Public Health
Agency (PHA), Evidence Research and Innovation (ERI), Strategy and Policy, Maori Health and Maori
Crown relations, Regulations, System Performance and Monitoring, Government and Executive
Services, Corporate Services and Clinical leadership)
Key take home:
• Science Technical and Advisory (STA) team will stay at the MoH within PHA directory
• PHA includes: the Intelligence, Surveil ance and Knowledge team (this is made up of STA,
Intel igence and analytics and some broader communicable disease teams).
• The ERI may also be of interest to TAG members. This is a completely new directorate which
will working with STA on many pieces of advice.
10.0
Next Steps/Decisions Pending
Monkeypox update:
• There has of yet been no probable or confirmed case of Monkeypox in New Zealand, as of the
current date (8th July).
1982
• A test definition has been agreed on and monkeypox has been made a notifiable disease.
• The advice provided by this TAG was to use vaccination as a strategic approach, targeting close
contacts post-exposure rather than a broader public health vaccination.
ACT
• New Zealand government is looking at options to secure the third-generation vaccine. Of note
the current stock is second-generation and expired
• There is a smal pool of stock of cidofovir. Trying to get access to tecovirimat, however this is
limited internationally.
• Work is underway to get access to vaccinia immune globulin and antivirals from Australia if
required. New Zealand is in the queue for next year, although exact quantities has been
requested.
INFORMATION
• A request for advice has been received around pre-exposure prophylaxis for specific groups
(i.e., MSM) and if this is relevant in a New Zealand context. Discussion with CV TAG members
suggested use post exposure for close contacts may be more appropriate (pending international
experience).
11.0
Closing Karakia
OFFICIAL
Meeting closed at
2:53 pm
THE
Next meeting
05 August 2022
UNDER
RELEASED
 1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
1982
ACT
INFORMATION
OFFICIAL
THE
UNDER
RELEASED





Document 12
Memo
Internal Review of COVID-19 Testing Technical Advisory Group (CT TAG) for
Purpose and Function
Date:
14 February 2022
To:
Dr Ashley Bloomfield, Director General of Health
1982
Copy to:
Dr Ian Town, Chief Science Advisor
Bridget White, Deputy Chief Executive, COVID-19 Health System Response
ACT
Gil Hal , Group Manager, Science and Insights
Darryl Carpenter, Group Manager COVID-19 Testing and Supply
Dr Dan Bernal, Manager, Scientific and Technical Advisory
From:
Kirsten Beynon, Chief Testing Advisor
INFORMATION
For your:
Decision
Purpose of report
1.
The purpose of this memo is to outline the initial findin
OFFICIAL gs of the internal review of the
COVID-19 Testing Technical Advisory Group (CT TAG). In particular, this review has focused on
the purpose and function of the Group, and how this expertise can be best used in the future.
THE
Background
2.
In June 2021, Sir David Skegg as Chair of the Strategic Public Health Advisory Group,
UNDER
recommended to Hon Dr Ayesha Verral , Associate Minister of Health, that the government
establish an expert committee to advice on laboratory testing issues and ongoing strategy
that should arise over the fol owing eighteen months.
3.
The CT TAG was established in October 2021 to provide rapid, independent, and practical
advice to the Director General of Health on testing technologies, to inform New Zealand’s
COVID-19 response a
RELEASED nd the work to reconnect Aotearoa New Zealand to the world
[refer to
HR 20211861].
4.
It was envisaged that the function of the CT TAG was to advise and give expert oversight on:
assessment of the benefits and limitations of new testing technologies and paradigms
for New Zealand, including the suitability of new technologies in different settings and
scenarios,
comparison of new technologies to those currently in use,
technical guidance on the expected timeframe for adopting a new technology,
horizon-scanning to identify technologies being developed local y and overseas, and
Page 1 of 5


Document 12
the application of new technologies to assist in reconnecting New Zealand, including
their use in border management, and managed isolation and quarantine settings.
5.
Members were appointed, with Professor David Murdoch as the Chair. The group initially
comprised of a Chair and members with expertise in relevant areas such as microbiology,
epidemiology, immunology, clinical diagnostic testing review and development,
operational implementation and management and primary care. In addition, the Ministry’s
Chief Science Advisor was also a member.
Internal Review
Review Process
1982
CT TAG work to date
ACT
6.
In October 2021, a sub-group of the CT TAG led by Professor Murdoch, undertook
A Rapid
Review of COVID-19 Testing in Aotearoa New Zealand, often referred to as the ‘Murdoch
Review.’ The group was also engaged in the development of some rapid pieces of work
including the endorsement of the minimum criterial for selection and approval of Point of
Care (POC) technologies and input into the
COVID-19 Testing Strategy for Aotearoa New
Zealand.
7.
The Murdoch review made a range of recommendations including to “strengthen the
INFORMATION
leadership capacity and capability for testing within the Ministry of Health…. a new
appointment with a strong leadership background in laboratory operations, diagnostic
testing and testing strategy.”1
8.
To meet this requirement within the Ministry of Health (the Ministry) I, Kirsten Beynon, was
appointed Chief Advisor Testing. In addition, the Murdoch review articulated a further 12
OFFICIAL
recommendations for the Ministry of Health (the Ministry), including developing
strategies, a focus on innovation, modelling, workforce, horizon scanning, regular
THE
assessment and review. The Ministry responded to these recommendations in a report to
Minister Verrall,
Response to the Rapid Review of COVID-19 Testing in Aotearoa New
Zealand [HR20212072 refers].
9.
CT TAG members were notified
UNDER by you, the Director General of Health, in an email dated
24 December 2021 that my appointment had brought us to a ‘natural point of review’ as
to the purpose of the group. It was indicated that there would be steps taken in the New
Year and that as Chief Advisor Testing, I would lead engagement with the group on this
matter.
10.
To date, I have engaged with both the CT TAG and members of the group, on a range of
RELEASED
urgent and emerging issues, including the rapid revision of the minimum criteria for
selection and approval of POC technology and on a population based targeted testing
approach for PCR to optimise laboratory capacity. I have also engaged the New Zealand
Microbiology Network (NZMN) on a range of topics when wider Clinical Microbiologist
expertise and opinion has been required. These engagements have highlighted the
chal enges of and need to, pivot the purpose, function and focus of the CT TAG to ensure
we use their advice and expertise in a way that is most beneficial to New Zealanders.
1 COVID-19 Testing Technical Advisory Group – Sir David Murdoch,
A Rapid Review of COVID-19 Testing in Aotearoa New Zealand, 4 October
2021, page 4.
Page 2 of 5


Document 12
Initial Findings
11.
Since my appointment, the CT TAG has been convened on a ‘as required’ basis to support
decision making. This was managed predominantly through email engagement to support
rapid input into reports.
12.
It is clear that within the context of both the Delta and Omicron outbreaks, and
subsequently the requirement to rapidly reassess the national testing response, the CT
TAG have been essential. They have provided independent expert opinion and advice, in
particular on POC testing, Rapid Antigen Tests (RATs), and the international supply chain
chal enges.
13.
Informed by discussions, the CT TAG members must be in an environment where they ca
1982n
speak freely to provide advice for consideration, rather than comment on completed
papers and decisions.
ACT
Proposed changes to CT TAG
Purpose and function
14.
COVID-19 Testing is, wil remain, a core pil ar of any pandemic response strategy. The need
for expert advice on the use and type of testing as we move forward, wil become even
more important as begin move through the phases of the Omicron Strategy and look to
INFORMATION
reconnect New Zealanders with the world.
15.
Horizon scanning, equitable testing approaches, advising on laboratory capacity, role of
surveil ance testing and seroprevalence, managing the ongoing chal enge of RATs and
potential for the introduction of new pathogens with relaxed border controls, are areas
that we are aware wil require the expertise of this g
OFFICIAL roup. By pivoting the purpose and
focus of the CT TAG, we aim to respond to some of these chal enges.
16.
From discussions and observations, it
THE is my view that the use of the CT TAG scientific
expertise and industry knowledge is better achieved through a proactive planning and
engagement approach, compared to the rapid reactive advice seeking methods.
17.
I recommend that the CT TAG is repurposed as a testing advisory group with a focus on
UNDER
industry intelligence providing advice on al facets of the COVID-19 testing system. I also
recommend that the membership and terms of reference are reviewed.
18.
The group should shift to a fortnightly meeting schedule, to be fully engaged in the
provision of advice, is forward-thinking. It is envisaged these meetings are less formal than
they have been, unless a specific decision is required from the group.
RELEASED
19.
Three areas which we are aware wil require specific consideration and input are a formal
review of testing operations, re-evaluation of POC RAT tests, and advance planning for
testing plan in phase 3 and beyond of the Omicron Strategy.
20.
As Ministry representative on the group, I propose to provide regular updates to you and
the Chief Science Advisor fol owing each meeting.
Membership
21. I recommend that:
to best align membership moving forward with a new purpose and function, we
consider including additional expertise and perspectives to the group,
Page 3 of 5




Document 12
we fill the role of scientific expertise on the group (currently vacant), ideally an
individual with a doctorate in laboratory science,
we seek additional members who will bring a strong equity and community
perspective, and
we consolidate attendees from the Ministry of Health.
22. If the purpose and function is agreed in principle, we wil provide further advice on
membership, reflective of the expertise required; noting that may not include some current
members.
Next Steps
1982
23. Following your ‘in-principle’ agreement to the changes outlined above, these wil be
ACT
discussed with the current members of the Group to ensure clarity of purpose and function
moving forward and seek their feedback.
24. A final paper seeking your agreement to update the CT TAG membership and terms of
reference, wil be provided to you in early March 2022.
INFORMATION
OFFICIAL
THE
UNDER
RELEASED
Page 4 of 5

Document 12
Recommendations
It is recommended that you:
1. Agree
The purpose of the CT TAG is shifted to a focus on considering
Yes/No
industry intelligence and providing advice on al facets of the
COVID-19 testing system.
2. Agree
The function of the CT TAG shifting to a fortnightly meeting with a
Yes/No
forward thinking, proactive approach to advice.
3. Note
If agreed, in principle, we will consult current members on the
Noted
1982
proposed changes.
ACT
4. Note
We will update you on proposed membership and revised terms
Noted
of reference and revised terms of reference in early March 2022.
Electronic
Signature ___________________________________________________
Date:
Kirsten Beynon
INFORMATION
Chief Testing Advisor
COVID-19 Health System Response
OFFICIAL
THE
Signature ___________________________________________________
Date:
Dr Ashley Bloomfield
Director General of Health
UNDER
RELEASED
Page 5 of 5













































