
 Assess Cannabis-Based Medicine and Product Funding
Assess Cannabis-Based Medicine and Product Funding
Requests
v9.0
Summary
Objective
To ensure recovery team members consistently access Recovery Support in order to make accurate decisions on funding requests
for cannabis-based medicine or products. In all cases written advice from a Pharmaceutical Advisor is necessary.
Background
There is increasing demand for ACC to fund cannabis-based medicines and products, and there needs to be a consistent process for
recovery team members to follow.
Owner
Outside of Scope
Expert
Outside of Scope
Procedure
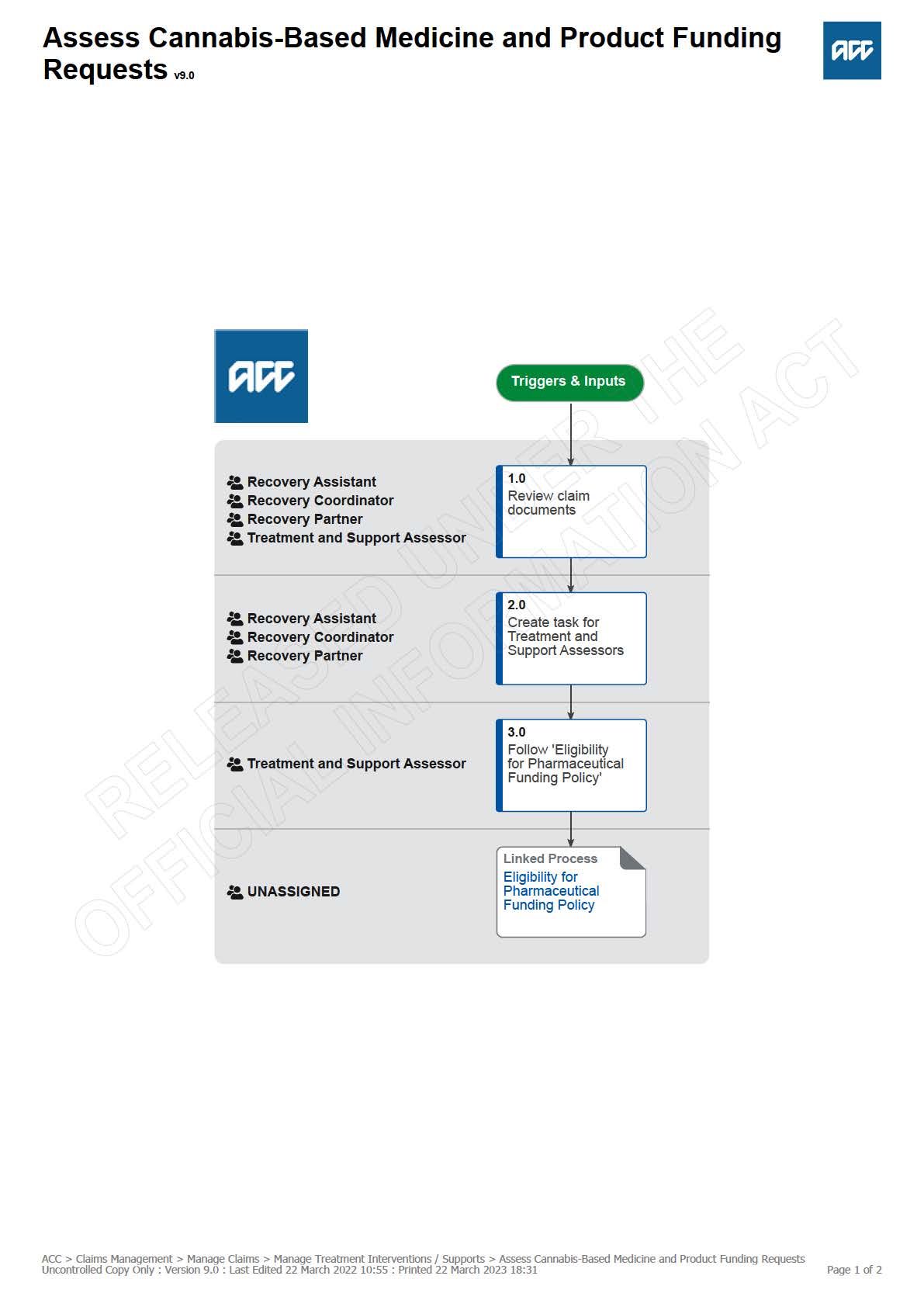
1.0 Review claim documents
Recovery Assistant, Recovery Coordinator, Recovery Partner, Treatment and Support Assessor
a Establish that there is a written request to fund a cannabis-based medicine or product.
NOTE What do you need to consider when the entitlement request is received and deemed cover exists?
Refer to the Deemed Cover and Entitlements Policy for considerations to determine client entitlement eligibility while
in deemed cover period.
Deemed Cover and Entitlements Policy
NOTE Which medicines or products are cannabis-based?
Current examples include Sativex, Tilray, Endocan, and Swift. However, there are many more cannabis-based prod-
ucts coming onto the NZ market. Some of these products may also contain THC (tetrahydrocannabinol; the principal
psychoactive component of cannabis).
b Ensure that the prescriber has completed an ACC1171 - Request for pharmaceutical funding.
ACC1171 Request for pharmaceutical funding form
NOTE What if the prescriber has not completed this form?
Check whether the ACC1171 is on file elsewhere. If so, proceed to 1c. If not, send the requestor the ACC1171 tem-
plate and ask that they complete this and return the form in order for the request to be considered.
c Ensure that part C of the ACC1171 form has been completed by a medical specialist who is overseeing prescribing.
NOTE What if this section is incomplete, or has been completed by someone other than the medical specialist over-
seeing prescribing?
Send the form back to the prescriber and request that part C is completed within 20 working days. If no response is re-
ceived within this timeframe, inform the provider that the request cannot be considered without the required infor-
mation. If no further information is provided at that time, decline the request.
Service Contracts and Contracted Providers - MFP spreadsheet
d Collate relevant clinical documents, such as any up to date clinical reports and/or GP notes, and social rehabilitation or needs
assessment reports, showing clinical and rehabilitation outcomes to date.
2.0 Create task for Treatment and Support Assessors
Recovery Assistant, Recovery Coordinator, Recovery Partner
a Create an "Alert: You Have Mail" task, which can be made at the ACC45 level.
b Label the task "ACC1171 Cannabis request attn T and S Assessor".
c Send the task to the "Elective SC - Claimant Reimbursements" queue.
3.0 Follow 'Eligibility for Pharmaceutical Funding Policy'
Treatment and Support Assessor
PROCESS
Eligibility for Pharmaceutical Funding Policy
UNASSIGNED
ACC > Claims Management > Manage Claims > Manage Treatment Interventions / Supports > Assess Cannabis-Based Medicine and Product Funding Requests
Uncontrolled Copy Only : Version 9.0 : Last Edited 22 March 2022 10:55 : Printed 22 March 2023 18:31
Page 2 of 2
 Eligibility for Pharmaceutical Funding Policy v11.0
Summary
Objective
Eligibility for Pharmaceutical Funding Policy v11.0
Summary
Objective
Pharmaceuticals are an entitlement ACC is liable to fund where necessary and appropriate to aid the treatment of a client’s personal
injury. See Schedule 1, Clauses 1-3 of the Accident Compensation Act 2001.
Pharmaceuticals are defined in Section 6 of the Act as:
(a) a prescription medicine, a restricted medicine, or a pharmacy-only medicine, as listed in Parts 1, 2, and 3 of Schedule 1 of the
Medicines Regulations 1984; or
(b) a controlled drug as defined in the Misuse of Drugs Act 1975.
Pharmaceuticals do not include vitamins, supplements, lollies, or other medical consumables. Instead see:
• Medical consumables policy
Owner
Outside of Scope
Expert
Outside of Scope
Policy
1.0 What do we fund?
a Pharmaceutical costs can be approved for payment in advance or reimbursed for costs already incurred. If we agree to pay the
cost of a pharmaceutical reimbursement or invoice, we pay:
• the prescription co-payment charge for fully-subsidised items.
• a contribution to partly subsidised and non-subsidised pharmaceuticals.
Subsidised pharmaceutical reimbursement costs
2.0 Who can ask for funding?
a Where the cost is for reimbursement of pharmacy co-payment costs already incurred, a client can request reimbursement (via
MyACC or ACC249 Prescription reimbursement form). These costs cover the prescription co-payment for subsidised items
only.
ACC249 Request for reimbursement of pharmaceutical costs
b Where the cost is for a partly subsidised or non-subsidised prescription, a treatment provider can request approval for funding
(see ACC1171 Request for pharmaceutical funding form).
ACC1171 Request for pharmaceutical funding form
c An approved pharmacy may direct bill ACC.
Process a manual vendor request
d The following treatment providers have statutory authority to prescribe pharmaceuticals within their scope of practice, and can
therefore request funding on behalf of a client for partly subsidised or non-subsidised pharmaceuticals:
• Medical practitioners and Medical specialists – including unapproved medicines or unapproved medicine use. Note that all
other prescribers cannot prescribe unapproved medicines.
• Dentists
• Nurse practitioners
• Nurse prescribers
• Pharmacist prescribers
• Optometrists
Treatment Providers with Prescribing Rights
3.0 Approval Stage 1: Criteria for subsidised pharmaceuticals
a Requests for all subsidised pharmaceuticals can be approved and reimbursed only where ALL of the following bullet points are
met:
• The client has cover accepted for a personal injury.
• The request covers pharmacy co-payments of fully subsidised pharmaceuticals only, so each item on the request is $18 or
less.
• There is a pharmacy receipt attached if the total schedule amount requested is over $50 (eg if multiple fully subsidised
pharmaceuticals are requested).
• The client has not made more than three requests under $50 in a week.
• The date of injury is stated to be within 12 months of the date of receipt or 24 months of the date of receipt or the date the re-
quest is made if it is a renewal.
NOTE What if any of the criteria are not met or you are not sure?
If any one of these criteria are not met, or you are not sure if it is met, then the request is referred to Treatment & Sup-
port and assessed in line with Approval Stage 2.
ACC > Claims Management > Manage Claims > Operational Policies > Treatment and Rehabilitation > Pharmaceuticals & medical consumables > Eligibility for Pharmaceutical
Funding Policy
Uncontrolled Copy Only : Version 11.0 : Last Edited Thursday, February 2, 2023 2:02 PM : Printed 22 March 2023 18:30
Page 1 of 4
NOTE As a Recovery Partner which Payments queue do you send the request to?
The Payments Team queue is ESC- Claimant reimbursement for client reimbursements.
Pharmacy direct billing should be sent to ESC-Pharmaceuticals.
NOTE As a Recovery Team Member which task type do you use to refer to ESC- Claimant Reimbursement Queue?
• For all tasks including ACC1171s, use an ‘Alert: You Have Mail’ task.
• For ACC1171s, ensure appropriate documentation is linked to the task and advise to ‘process as per attached’.
• An alternative task type for non-ACC1171s is the ‘Make MFP Payment’ task is fine.
Both types of task can be created from the ACC45 claim level.
NOTE How do you know if an item is a pharmaceutical?
To find out if an item is a pharmaceutical, search the item in the DataSheets and Consumer Medicine Information tab
on the Medsafe website. https://www.medsafe.govt.nz/Medicines/infoSearch.asp
Note that Pharmac's Pharmaceutical Schedule will tell you if an item is subsidised, but not whether it is a pharma-
ceutical. Pharmac subsidises both pharmaceutical and non-pharmaceutical items.
If you are not sure whether an item is a pharmaceutical or not send the request to Treatment and Support for Approval
Stage 2.
NOTE Are cannabis products eligible for funding under Stage One?
To clarify, no cannabis product is eligible for funding under Stage One as they do not meet the Stage One criteria.
NOTE How much is a fully subsidised pharmaceutical?
Fully subsidised pharmaceuticals are $5 for standard prescriptions and $15 for specialist prescriptions. We allow for
items $18 and under to be paid as if they are fully subsidised. Where any item on the request is over $18, the whole
request needs to be sent to Treatment and Support for Approval Stage 2.
NOTE What information does a receipt require?
Receipts are only required for requests over $50. The receipt must include: The client’s name, prescription number,
the prescribing treatment provider’s name, the date the pharmaceutical was dispensed (which must be on or after the
date of injury), the pharmaceutical name, the pharmaceutical cost, and any extra charges.
NOTE What if you are not sure how many requests under $50 a client has made in the last week?
If you don't have evidence of multiple requests from a client on hand, you do not need to follow this up. This step will
become automated in future. At this stage, this is a prompt to keep in mind that clients may be submitting multiple re-
quests without a receipt. Where you do notice this pattern, send the request to Treatment & Support for further
assessment.
b Where a client has reached reimbursements of $300 between 1 Feb to 31 Jan each year, you should still reimburse them, but
also send the request to Treatment and Support to alert the client to the Prescription Subsidy Scheme.
NOTE Why do we check that a client has not exceeded $300 of pharmaceutical reimbursements annually?
After receiving 20 prescriptions between 1 Feb and 31 Jan in a single year, a person becomes entitled to the Ministry
of Health’s Prescription Subsidy Scheme, meaning they no longer have to make co-payments for their pharma-
ceuticals. Because some co-payments can be $15 each for specialist scripts, our threshold is $300 before we check
whether the client should be receiving the Prescription Subsidy. If a client has paid a co-payment in these circums-
tances, we will still reimburse them but they should be alerted to the Prescription Subsidy Scheme.
Pharmac - Pharmaceutical Schedule
https://pharmac.govt.nz/pharmaceutical-schedule/community-section-b/
Eligibility for funding Pharmac subsidised pharmaceuticals
4.0 Approval Stage 2: Criteria for non-subsidised and partially subsidised pharmaceuticals - to be as-
sessed by Treatment & Support
a Requests for all non-subsidised and partially subsidised pharmaceuticals, and requests that do not meet the Approval Stage 1
criteria, can be approved and reimbursed only where ALL of the following bullet points are met:
• The item is a pharmaceutical
• The pharmaceutical is prescribed for a covered personal injury.
• The pharmaceutical is prescribed by a treatment provider with statutory prescribing rights.
• The pharmaceutical is approved in New Zealand for the covered condition (ie Medsafe approved with the right indication).
• At least one subsidised pharmaceutical with the same therapeutic effect (if the pharmaceutical is not subsidised already) has
been tried and failed or stopped due to side effects previously or there is no relevant alternative.
NOTE What if there are multiple items on the request and one or more of the items are not regulated?
Where there are multiple items on a request and one or more items are not regulated, the whole request needs further
assessment from Pharmaceutical Advisors in line with Approval Stage 3.
NOTE How will you know that a pharmaceutical is approved with the right indication?
You can search Medsafe's approval database by writing in the pharmaceuticals name and seeing whether a datasheet
exists. On the datasheet, look to see what conditions the pharmaceutical is indicated for, and in what form and dose it
is indicated in. If you have any questions, call the Pharmaceutical Advisors hotline.
ACC > Claims Management > Manage Claims > Operational Policies > Treatment and Rehabilitation > Pharmaceuticals & medical consumables > Eligibility for Pharmaceutical
Funding Policy
Uncontrolled Copy Only : Version 11.0 : Last Edited Thursday, February 2, 2023 2:02 PM : Printed 22 March 2023 18:30
Page 2 of 4
NOTE Are cannabis products eligible for funding under Stage Two?
To clarify, there is only one cannabis product that could potentially be funded under Stage 2. This product is Sativex.
Sativex is non-subsidised, but is registered by Medsafe. Sativex has a very narrow registered indication - spasticity
due to Multiple Sclerosis. Since the registered indication for Sativex is so narrow, it is highly unlikely that any cannabis
product will be eligible for funding under Stage Two criteria. Therefore, seek written guidance from a Pharmaceutical
Advisor so that they can consider any cannabis product request under Stage Three criteria.
Database of Medicine Classifications (medsafe.govt.nz)
https://medsafe.govt.nz/profs/class/classintro.asp
b Where there are multiple items on a request and one or more items are not regulated, the whole request needs further assess-
ment from Pharmaceutical Advisors in line with Approval Stage 3.
NOTE How do you know if a subsidised version of a pharmaceutical has been tried?
ACC1171 should contain this information. Engage with the prescriber for more information about what alternatives
have been tried and why a non-subsidised pharmaceutical is being prescribed now. To check whether a pharma-
ceutical is subsidised, check Pharmac's Pharmaceutical Schedule. If you need more assistance, call the Pharma-
ceutical Advisor hotline.
Pharmac - Pharmaceutical Schedule
https://pharmac.govt.nz/pharmaceutical-schedule/community-section-b/
c Where a renewal for a partly subsidised or non-subsidised pharmaceutical has been prescribed, you may not need to reassess
the request if:
• the same request and cost contribution has already been approved for the client and the renewal is a repeat of that exact re-
quest
• there were no specific criteria for the initial approval that would require reassessment against those criteria
• it is within three months of the date the pharmaceuticals were prescribed.
NOTE How do you know if you need written guidance or hotline guidance
All requests that include at least one unapproved medicine need Pharmaceutical Advisor Written Guidance. All other
support can be received on the hotline.
NOTE Why do renewals not need to be reassessed?
If we have already approved the same request before, under usual circumstances we should approve it again. This in-
cludes renewals where the request originally required Pharmaceutical Advisor Guidance. However, if there is an indi-
cation that the original request was only for a specific time period (eg three months) then the renewal will need to be
reassessed in line with the relevant criteria. A general rule of thumb for non-subsidized pharmaceuticals is to consider
approval for up to but no greater than a period of 12 months.
d Where a client has exceeded reimbursements of $300 between 1 Feb to 31 Jan each year, alert the client to Ministry of
Health's Prescription Subsidy Scheme which means they no longer have to make co-payments for their pharmaceuticals.
e If the pharmaceutical is not prescribed for a covered injury by a treatment provider with statutory prescribing rights, decline the
request. If the other criteria are not met, or you are unsure, then the request is referred to Pharmaceutical Advisors and as-
sessed in line with Approval Stage 3.
Seek Internal Guidance
Eligibility for funding under non-subsidised pharmaceuticals
5.0 Approval Stage 3: Criteria for non-regulated pharmaceuticals - to be assessed by a Pharmaceutical Ad-
visor
a If the request is for a pharmaceutical that is not regulated by Medsafe for the covered condition, or criteria from Approval Stage
1 and 2 are not met, the balancing of a number of factors needs to be considered before you can make a decision to approve
or decline the pharmaceutical funding.
b The answers to these questions should be indicated by the prescribing medical practitioner in their referral, and clinically as-
sessed by you:
• Have all other options been exhausted?
• Is the pharmaceutical regulated for the covered condition in another jurisdiction, eg by the Therapeutic Goods Administration
(Australia) or the Food and Drug Administration (USA)?
• What available research on safety and efficacy is there and what is the strength of that research?
• What is the prescribing medical practitioner’s rationale for prescribing the pharmaceutical? What is the clinically assessed
potential rehabilitation outcome?
• Is there specialist medical practitioner support involved for unapproved medicines use?
• What is the cost in comparison to alternative or similar treatment options, if they exist?
c Where this information has not been provided, you need to seek more information before declining the request.
d Consider whether confirmation from an external specialist is appropriate to inform or confirm the decision. External opinion will
at least carry weight equal to the prescribing practitioner, and will depend on the qualifications and scope of practice (as dic-
tated by the registering body) the prescribing/applying specialist holds.
NOTE How will you know if an external specialist opinion is needed?
An external specialist opinion will usually only be required where there are conflicting clinical opinions, or the re-
quested pharmaceutical or its use in this condition is particularly innovative or novel.
ACC > Claims Management > Manage Claims > Operational Policies > Treatment and Rehabilitation > Pharmaceuticals & medical consumables > Eligibility for Pharmaceutical
Funding Policy
Uncontrolled Copy Only : Version 11.0 : Last Edited Thursday, February 2, 2023 2:02 PM : Printed 22 March 2023 18:30
Page 3 of 4
 e
e Where the request is for a non-regulated oncology pharmaceutical or an oncology pharmaceutical that is not approved to treat
the specific condition, after making a recommendation to seek more information or to decline the funding, seek further guid-
ance from a Clinical Advice Manager.
ACC > Claims Management > Manage Claims > Operational Policies > Treatment and Rehabilitation > Pharmaceuticals & medical consumables > Eligibility for Pharmaceutical
Funding Policy
Uncontrolled Copy Only : Version 11.0 : Last Edited Thursday, February 2, 2023 2:02 PM : Printed 22 March 2023 18:30
Page 4 of 4
Document Outline
- Assess Cannabis-Based Medicine and Product Funding Requests
- Eligibility for Pharmaceutical Funding Policy




