Rev Environ Health 2016; 31(3): 329–352
Jackie Wright*, John Edwards and Stewart Walker
Exposures associated with clandestine
methamphetamine drug laboratories in
Australia
DOI 10.1515/reveh-2016-0017
Introduction
Received April 20, 2016; accepted June 7, 2016; previously published
online July 18, 2016
Illicit drugs such as amphetamine-type stimulants (ATS)
Abstract: The clandestine manufacture of methamphet- (1) are manufactured in Australia within clandestine
amine in residential homes may represent significant laboratories that range from crude, makeshift operations
hazards and exposures not only to those involved in the using simple processes to sophisticated operations. These
manufacture of the drugs but also to others living in the laboratories use a range of chemical precursors to manu-
home (including children), neighbours and first respond- facture or “cook” ATS that include methylamphetamine,
ers to the premises. These hazards are associated with more commonly referred to as methamphetamine (“ice”)
the nature and improper storage and use of precursor and 3,4-methylenedioxymethamphetamine (MDMA or
chemicals, intermediate chemicals and wastes, gases and “ecstasy”). In Australia the primary ATS manufactured
methamphetamine residues generated during manufac- in clandestine drug laboratories is methamphetamine
ture and the drugs themselves. Many of these compounds (2), which is the primary focus of this review. Clandes-
are persistent and result in exposures inside a home not tine laboratories are commonly located within residential
only during manufacture but after the laboratory has been homes, units, hotel rooms, backyard sheds and cars, with
seized or removed. Hence new occupants of buildings for- increasing numbers detected in Australia each year (744
merly used to manufacture methamphetamine may be laboratories detected in 2013–2014) (2). Unlike the legal
unknowingly exposed to these hazards. Children are most manufacture of industrial and pharmaceutical chemicals,
susceptible to these hazards and evidence is available in clandestine drug operations do not involve any care in the
the literature to indicate that these exposures may result storage, handling and disposal of chemicals and wastes
in immediate and long-term adverse health effects. The nor any responsibilities in relation to health and safety
assessment of exposure within the home can be under- during and after the cook. Many of these laboratories are
taken by measuring contaminant levels or collecting within urban communities where there are significant
appropriate biological data from individuals exposed. To hazards (including chemical exposures) to cooks, other
gain a better understanding of the available data and key residents, neighbours, law enforcement and other first
issues associated with these approaches to the characteri- responders and the general public who may visit or reoc-
sation of exposure, a review of the published literature cupy the premises.
has been undertaken.
Environmental exposures to illicit ATS drugs and
Keywords: biological monitoring; clandestine drug manu- chemicals used to manufacture them are not well defined,
facture; drugs of abuse; exposure.
particularly for children. From its initial establishment
through its ultimate re-occupancy, a clandestine drug lab-
oratory typically goes through a number of phases where
there is the potential for environmental exposures to the
manufactured drug and a wide range of chemicals associ-
ated with the manufacture of these drugs. These phases
*Corresponding author: Jackie Wright, Health and Environment,
include (3):
School of the Environment, Flinders University, GPO Box 2100,
– An operational phase, with the potential for exposure
Adelaide, SA 5001, Australia, E-mai
l: [email address]
to a large number of chemicals including the manu-
John Edwards: Health and Environment, School of the Environment,
Flinders University, Adelaide, SA, Australia
factured drug.
Stewart Walker: Chemical and Physical Sciences, Flinders
– A discovery phase, where the lab is “seized” by police
University, Adelaide, SA, Australia
and chemicals and equipment are removed. Residents
Unauthenticated
Download Date | 10/11/16 7:20 AM
330 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
may remain on the premises, or return immediately that specifically provides a minimum term of 12 months
after police have completed their investigations, and of imprisonment for anyone who causes harm to a child
be exposed to a wide range of chemicals that remain through the manufacture of drugs (15). Outside of crimi-
in the premises.
nal offences specifically related to harm caused during the
– A post operation/discovery/remediation phase, manufacture of an illegal drug, the laws that relate to the
where exposures may be associated with a former protection of the health of the general public who may be
laboratory that was undetected (so not remediated); exposed to contamination in a former ATS drug laboratory
was a known laboratory but not remediated; or was a are enforced by local authorities including councils (13, 16,
known laboratory that has not been adequately reme- 17), and typically relate to “nuisance” issues or premises
diated. In these premises exposure can occur to per- not being in a safe or healthy condition (e.g. NSW Local
sistent chemical and drug residues inside and from Government Act 1993, Western Australian Health Act 2011,
dumped waste materials outside (4–6).
Victorian Public Health & Wellbeing Act 2008 and South
Australian Public Health Act 2011). These instruments (and
The greatest hazard, both in relation to likelihood of others) generally provide limited powers to prevent a prop-
exposure and concentrations that may be present, occurs erty being re-occupied prior to remediation.
during the operational phase. This is where the potential
Ultimately it is the role of the property owner to
for inhalation of airborne contaminants (including meth- ensure their property is suitable for occupation. Legisla-
amphetamine and gases that include acidic, corrosive and tion is available in various states that require a landlord to
toxic gases) and direct contact with primary chemicals, provide residential premises that are clean and fit for habi-
wastes and drug products, and the presence of physical tation (e.g. NSW Residential Tenancies Act 2010, Victorian
hazards that may be flammable, reactive of explosive may Residential Tenancies Act 1997, Queensland Residential
occur (7, 8). The clandestine manufacture of ATS places Tenancies and Rooming Accommodation Act 2008 and
several groups of people at risk including adults (such as South Australian Residential Tenancies Act 1995). Such
the drug “cooks”), children, neighbours, police, foren- legislation typically states that the tenant must not use
sic scientists and emergency workers (7, 9–11). Children the premises for any illegal activity or purpose.
living in proximity to clandestine laboratories operated by
To better understand the potential for exposure in
parents or family members are at increased risk of injury premises where ATS, specifically methamphetamine,
and adverse health effects (9, 12).
have been manufactured, this review has been under-
Australia has developed guidelines relating to the taken to identify the available information that relates to
assessment and remediation of contamination (3, 13) that characterising exposure within homes used to manufac-
include human health risk-based guidelines for indoor air, ture methamphetamine and adverse health effects.
indoor surfaces and outdoor environments in residential,
commercial and public open space areas (3). These guide-
lines consider physical assessment and remediation of
Background information on
property/premises formerly used for the manufacture of
ATS. However, there is limited guidance on assessing and
clandestine drug laboratories in
managing individual exposures and health risks (particu-
Australia
larly in children) during the operation of the laboratory,
immediately after seizure or if the property is not remedi-
ated and is re-occupied.
General
In Australia, the
Law and Justice Legislation Amend-
ment (
Serious Drug Offences and Other Measures)
Act 2005 ATS are a group of psychostimulant drugs that are related
[the SDO Act (14)] includes offences, that carry custodial to the parent compound, amphetamine, and have a wide
sentences, for endangering children during activities asso- range of common/street names (18). The manufacture of
ciated with the manufacture of controlled drugs or precur- methamphetamine involves a relatively simple chemi-
sors. Most Australian state legislation and initiatives focus cal processes that use highly flammable, very toxic and
on penalties and harm reduction measures associated with corrosive chemicals (7). The first clandestine ATS labora-
drug use, possession and trafficking, with some provisions tories were found in San Francisco and the surrounding
for offences that relate to manufacture, or equipment or Bay area around 1962 with the first Australian clandes-
precursors used for manufacture of drugs (7). One state, tine ATS laboratory reported to be in Sydney in 1976 (19).
Western Australia, has introduced stronger legislation The number of clandestine drug laboratories detected in
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
331
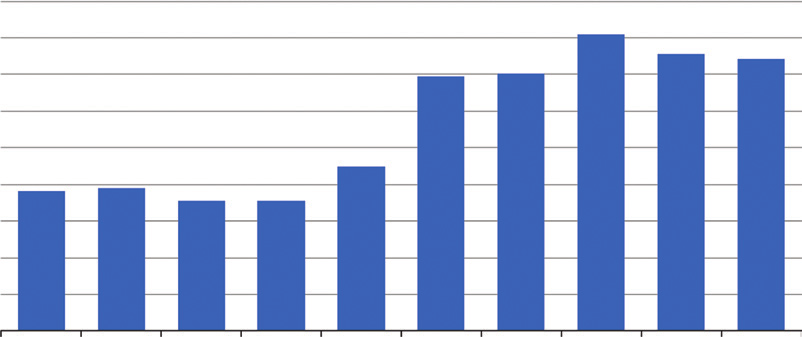
Australia have since increased year-on-year with numbers
of detections over the past decade shown in Figure 1. The
number may be higher than this as data from New Zealand
indicates that 32% of frequent drug users in 2011 indicated
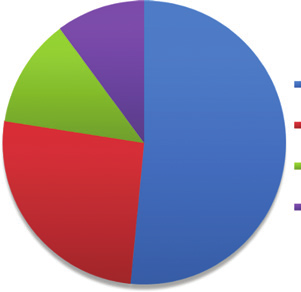
Addict-based labs (51.6%)
that they cooked (or had an attempt at cooking) their own
Other small-scale labs (26.0%)
drugs (20). It is estimated that approximately only 1 in 10
laboratories are detected in Australia (21).
Medium sized labs (12.3%)
The Internet contributes to local methamphetamine
Industrial scale labs (10.1%)
production due to the increased ease of access to chemi-
cal precursors, equipment and information (9). Scales of
clandestine drug manufacture range from easily transport-
able small-scale ‘boot labs’ (so-called because they can fit
into the boot of a car for easy transportation) and smaller
Figure 2: Size and production capacity of clandestine drug labora-
tories detected in Australia in 2013/2014 (2).
addict-based laboratories to more permanent large-scale
laboratories (22) with the distribution of different sized
laboratories detected in 2013–2014 illustrated in Figure 2.
From 2008 to 2013 between 68% and 71% of the clan- Drug Laboratory Remediation Guidelines (13). Of the clan-
destine laboratories in Australia were detected in residen- destine laboratories detected in 2013–2014 (2) 78.9% were
tial areas with the rest from commercial/industrial, rural associated with the manufacture of ATS with < 1% asso-
areas and vehicles (1, 2, 18, 23–25). The increasing detec- ciated with the extraction of precursor chemicals pseu-
tion rate of clandestine laboratories, particularly in urban doephedrine and ephedrine. Most of the ATS laboratories
residential areas in Australia, has resulted in an increase seized, (99%) were associated with the manufacture of
in media reports, particularly in relation to injuries and methamphetamine and amphetamine, with the remainder
public risks associated with explosions, exposures by associated with the production of MDMA.
police during seizures, the presence of children at these
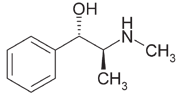
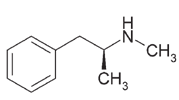
Pseudoephedrine is the preferred primary precursor
premises and general community concerns.
for the manufacture of methamphetamine due to the ease
of conversion (21), where the reaction required involves
the removal of a single hydroxyl group from the pseu-
Drugs manufactured and common methods
doephedrine molecule to produce methamphetamine
(refer to Figure 3) (21).
Since the late 1970s over 100 “recipes” or methods used to
There are four main methamphetamine manufactur-
manufacture ATS have been identified by the Australian ing methods that have been identified in Australia (1–3, 7,
Crime Commission (3) in support of the national Clandestine 19, 23–25) with clear geographic distributions:
900
800
700
600
500
400
Number
300
200
100
0
2004–05
2005–06
2006–07
2007–08
2008–09
2009–10
2010–11
2011–12
2012–13
2013–14
Figure 1: Number of clandestine drug laboratory detections in Australia: 2005/05–2013/14 (2).
Unauthenticated
Download Date | 10/11/16 7:20 AM
 332
332 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
manufacture of methamphetamine that include the use of
corrosive, explosive, flammable and toxic chemicals (5, 12,
26–32).
More generally, the manufacturing of metham-
Pseudoephedrine
Methamphetamine
phetamine from ephedrine and pseudoephedrine (most
common and preferred method in Australia) has the
Figure 3: Reduction of pseudoephedrine to methamphetamine.
potential to result in contamination from the storage and
use of precursors and chemicals, gases released during
– Hypophosphorous (or Hypo) method (which is a various stages of manufacture, methamphetamine resi-
variation of the red phosphorous method) where dues and waste materials.
ephedrine or pseudoephedrine, iodine and hypophos-
Use and storage of precursors and chemicals: The
phorous acid are used. This is the most common collection, often illegal (33), and storage precursor chemi-
method of methamphetamine manufacture in Aus- cals including (1) cold and allergy medications, drain
tralia accounting for approximately 63% of identified cleaner, rock salt, battery acid, lithium batteries, pool
ATS laboratories in 2013–2014, primarily in the east- chloride, iodine, lighter fluid, matches, fireworks, distress
ern states (2, 21).
flares, antifreeze, propane and paint thinner. Waste mate-
– Ammonia (“Birch” or “Nazi”) method where ephed- rials may also be stored within the premises. Given the
rine or pseudoephedrine is reduced in a chemical illegal nature of the manufacturing process these chemi-
process involving anhydrous ammonia and lithium cals are often stored in unlabelled and unsuitable contain-
or sodium metal. Despite the hazards associated with ers (including containers with no lids or food containers)
this method, it is quick and efficient (21) and accounts that result in accidental ingestion (34) or leaks and spills;
for approximately 21% of the identified ATS labora- or dumped into drains, soil or waterways (29, 35). Precur-
tories in 2013–2014, principally in Western Australia sor chemicals have been found at high concentrations
(2, 18).
in kitchen appliances such as microwaves (36), where
– Red phosphorous (or Red P method) method where contamination of food items prepared in these areas can
ephedrine or pseudoephedrine is reduced using occur. Methamphetamine has been detected in chicken
red phosphorous (extracted from match box striker removed from a refrigerator where it was adjacent to a jar
plates) and hydriodic acid. This method accounted of methamphetamine solution (37).
for approximately 7% of identified ATS laboratories in
Chemicals used in the manufacture of methampheta-
2013–2014, primarily in the eastern states (2).
mine include volatile solvents (8, 27, 34) that result in direct
– Phenyl-2-propanone (P2P) method (not common in irritation, inhalation exposures and systemic absorption.
Australia), using either the Leuckart method or the
Gases released during manufacture: Cooks using
“Hells Angels’” method where P2P is reduced using the ammonia method readily produce ammonia gas (38).
formamide, ammonium formate, formic acid, methyl- Cooks using the red phosphorous and hypophospho-
amine, mercuric chloride, aluminium foil and metha- rous methods produce phosphine gas (39). Both of these
nol. This method accounted for approximately 4.5% gases are toxic and in enclosed spaces, can reach high
of identified ATS laboratories in 2013–2014, primarily concentrations resulting in direct irritation and inhala-
in the eastern states (2).
tion exposures/systemic absorption and injuries (40, 41).
Phosphine in particular has poor odour warning proper-
ties and unwitting fatal exposures have been reported
Activities that give rise to contamination and (42). Hence bystanders and neighbours may recognise
exposure pathways
some ‘chemical odours’ such as pungent ammonia yet
may not notice other more harmful gases or vapours.
During the manufacture of methamphetamine, a range of
Gases that are produced during the cooking process
chemicals are used as precursors, produced as by-prod- are absorbed into porous materials and may off-gas over
ucts, and drug products may be present in air as vola- time resulting in inhalation exposures after the cook has
tiles or gases, deposit on surfaces within the home or be been completed. Limited data are available on this off-
present in liquid waste that may be dumped down drains, gassing process, its duration and its role with respect to
stored in various containers indoors or dumped outside exposure and health risks.
(to soil or water) (3). There are many general reviews that
Release of iodine residues: Iodine is released (27)
identify a range of chemical hazards associated with the during the manufacturing process (red phosphorous and
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
333
hypophosphorous methods) and forms a surface residue distribution of methamphetamine residues from smoking
that often stains the walls of a room where the drug was (48).
manufactured. These surface residues can result in expo-
The initial product of methamphetamine synthesis is
sures via dermal absorption and ingestion following the free base form of the drug, which is volatile and does
transfer to hands and objects.
not persist in the environment for any significant period
Methamphetamine residues: Methamphetamine is of time (49). The hydrochloride salt is persistent in the
generally produced as the free base or the hydrochloride environment, although its stability is pH dependant (49).
salt. Methamphetamine base is an insoluble oil at room At a pH in excess of 4 or 5, the hydrochloride salt is more
temperature and is the first product of illegal manu- unstable and the more volatile free base is formed (49).
facture. It is not suitable for injecting and is difficult to
Activity in a residence where methamphetamine has
snort (43). Hence it is converted to its hydrochloride salt, been manufactured can result in re-suspension of respir-
usually by bubbling hydrogen chloride gas through an able fractions resulting in the potential for ongoing inha-
alcohol or diethyl ether solution of methamphetamine lation exposures (50).
base (3, 8). This process is referred to as “salting out” is
Distribution of methamphetamine residues:
associated (44) with the release of respirable (predomi- Methamphetamine residues on wall surfaces increase in
nantly < 1.0 μm diameter) aerosols of methamphetamine concentration with height above the floor (51). It is not
(and hydrochloric acid) that can be directly inhaled clear if the distribution of methamphetamine residues
or transported throughout the premises and residues is solely due to the manufacture of the drug or if there
deposited on surfaces (hard and soft). Contaminants is a contribution from the occupants who also may have
present in these residues may be absorbed through the smoked the drug (common in the US where the study sites
skin (45, 46) or ingested (from placing hands or objects are located).
in the mouth).
Methamphetamine is absorbed into porous surfaces
Waste materials: It has been estimated that for each including concrete and paint on surfaces that include
kilogram of methamphetamine manufactured, 6–10 kg of gyprock walls (plasterboard or drywall) (52, 53) and
waste are produced (8) that is often dumped in drains or carpets (54). Elevated levels have been found in painted
outside, directly into the soil.
plasterboard surfaces (51, 55), with lower levels found in
In the event of a fire or explosion contamination from the plasterboard paper (front and back), and no detec-
precursors, intermediates, products, wastes and combus- tions within the gypsum itself (51).
tion products are more readily and rapidly spread through-
Methamphetamine adsorbed into gyprock walls
out the premises and to neighbouring homes. Emergency can desorb over time (depending on temperature and
personnel are potentially exposed to these contaminants humidity) contributing to ongoing exposures in a home
if not properly protected.
(52, 53).
Persistence: Without remediation, residues may
persist for months at least, and result in exposures and
Fate and transport of methamphetamine
contamination of clothing of all individuals who enter the
indoors
premises (36, 47, 50, 53, 56). An initial study (57) on the
persistence of methamphetamine residues on wall sur-
The fate and transport of methamphetamine indoors has faces over time has indicated a reduction of approximately
been studied more extensively than other chemical inter- 50%–60% after 47 days and up to 80% after 179 days
mediates, wastes and products. The behaviour of meth- (with no remediation). The persistence is expected to vary
amphetamine indoors has been determined from studies depending on a wide range of factors that include pH,
(including “controlled cooks”) where levels of metham- temperature and humidity.
phetamine on indoor surfaces and other materials have
Removal and remediation: It is suggested that
been measured.
washing of surfaces removes a significant portion of meth-
Release and transport of methamphetamine amphetamine surface residues, in particular dislodgeable
residues: Methamphetamine is released as an aerosol residues which would be re-suspended with activity in the
during the production process and transported in air to premises (55). Hence following initial cleaning of a premise
locations distant from the site of synthesis. Hence surface the potential for fine particles of methamphetamine that
residues associated with methamphetamine production can be re-suspended and inhaled is expected to be very low
are found throughout the premises not just in the room(s) and not expected to be of concern. There are, however, no
used for manufacture (36, 38, 39, 47) consistent with the published data to specifically support this outcome. Work
Unauthenticated
Download Date | 10/11/16 7:20 AM
334 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
in the United States (57) and South Australia (Edwards
Drug cooks
pers. comm.) suggests that some surface contamination is
easily removed, however, deeper contamination in porous Limited published data are available on drug cooks whose
materials (including surfaces such as plasterboard, con- exposure to physical hazards, precursor chemicals,
crete, plywood) can be more intractable and has required intermediates and wastes (including gases generated)
repeated attempts at washing, with and without detergents and methamphetamine during and after manufacture is
and/or bleaches, before surfaces have been tested and expected to be significant. Many cooks do not take basic
found to be effectively remediated. Data from New Zealand laboratory precautions such as wearing personal pro-
(55, 58) indicates that the washing is effective in reducing tective equipment (PPE) and have limited knowledge of
methamphetamine contamination of glass windows, is the consequences of mixing many of the chemicals, par-
partially effective for PVC, laminate or ceramic surfaces but ticularly in the presence of heat/open flames (27, 33). In
has no significant effect on wallpapered, painted or var- addition poor ventilation, common in illegal laboratories
nished surfaces. Stronger cleaners that contain oxidisers to avoid detection, increases the risk of exposure to high
(such as those that contain sodium hypochlorite or quater- concentrations of chemicals and by-products in air as well
nary ammonia) have been found to be more effective in the as fires and explosions (41, 42, 60). Given the illegal nature
cleaning of these surfaces (55). These cleaners have a very of the manufacturing operation no specific data are avail-
high pH, and given the pH-dependant stability of the more able in relation to the use of PPE.
persistent methamphetamine hydrochloride salt, their
A review of hospitalisation data from the US (61)
effectiveness is consistent with both the cleaning process showed that exposure of cooks resulting in injuries that
and potential conversion of the residue to the more volatile required hospitalisation were primarily from clandestine
base.
laboratories in their own residence with methampheta-
The efficacy of paint encapsulation in the remedia- mine, ammonia and hydrochloric acid the most com-
tion of methamphetamine residues on plasterboard has monly reported chemical exposures.
been found to depend on the type of paint used. Encap-
sulation with latex paint has not been shown to effectively
seal methamphetamine residues in place (51, 53, 55). Oil-
First-responders and forensic investigators
based paints have been found to be more effective with
the studies available indicating almost 100% still encap- First-responders (including police, fire fighters, ambu-
sulated 4 ½ months after painting (55).
lance and emergency personnel) are exposed to chemicals
Residues on porous clothing materials have been during discovery of clandestine laboratories in vehicles,
found (55) to be effectively removed with normal police raids on domestic or commercial premises or when
household washing, with a single standard wash removing fire fighters respond to a fire or explosion, or indirectly
more than 95% of methamphetamine contamination.
where these personnel treat contaminated and injured
individuals within or removed from the laboratory (11, 62).
Exposures by first-responders are higher during initial
Exposure issues associated with
entry into these premises, often when the presence of the
methamphetamine laboratories
laboratory is unknown (11), compared with exposures
that may occur in areas outside of, and adjacent to, the
laboratory.
General
Acute effects have been published, primarily from
the US, by police, fire fighters and investigators at seized
Anyone involved in the manufacture of methampheta- methamphetamine laboratories (40, 63), with a 7–15 fold
mine, or who accesses the premises used in its manufac- increased risk of illness reported (64). Adverse health
ture, has the potential to be exposed to physical hazards, effects and injuries in first-responders to unknown meth-
precursors, intermediates (including gases), waste prod- amphetamine laboratories (with or without fire or explo-
ucts and methamphetamine via inhalation, dermal sion) have been reported (29, 65) most commonly by police
absorption, ingestion and accidental injection (where officers (70%), emergency medical personnel (11%), fire-
users are also present). In addition approximately 20% of fighters (10%) and hospital personnel (9%). Chemicals
laboratories discovered in homes (59) result in explosions exposures most commonly reported by first responders
with severe injuries and exposures occurring within the in the US are derived from inhalation, with exposure to
premises and to neighbours.
ammonia and hydrochloric acid accounting for 54%–58%
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
335
of the injuries, and exposure levels to phosphine gas
Children do not have the same sense of danger as
reported well-above occupational limits (11, 42, 62). Other adults and will not understand the implications of playing
exposures may occur by skin contact and by touching with or near chemicals used in the manufacture of meth-
clothing of contaminated individuals removed from the amphetamine and will not be experienced with ways of
methamphetamine laboratory (11, 29, 40).
escaping from emergencies such as fires and explosions
The use of PPE by first-responders in the US is poorly (71).
reported and may be as low as 15% (11, 29, 60) with only
Between 25% and 40% (61, 74–77) of homes seized in
25% of personnel decontaminating at the scene (36). PPE the US were reported to have children present. The number
may be available on a planned raid of a clandestine labo- of children in these premises in the US has been observed
ratory, however, the level of chemical exposure is often to be increasing with the rate doubling between 1999
not known and the need for “speed and surprise” and the and 2002 (78). This may be due to the increased aware-
possibility of hostile actions and “booby-traps” (66) from ness of issues associated with exposures by children, and
occupants of the premises during the raid limit use of PPE increased reporting of children in these premises through
(63). Some guidance is available (64, 67) for emergency the introduction of Drug Endangered Children Programs
medical personnel in relation to the identification and in the US. Data from Australia are limited (8, 79), but
management of exposures in clandestine laboratories, anecdotal reports suggest children are commonly found
however, protocols adopted by various members of police, in clandestine drug laboratories and that these children
investigators, fire-fighters and medical staff are specific to have been exposed to chemicals and drugs present in
these organisations and may not consider these aspects.
these homes (7). Statements from children removed from
Once a laboratory has been seized exposures by those these premises (34) that indicate that drugs were often
involved in the further investigation of the site can still manufactured in the kitchen, with drugs and precursors
occur. These investigations include the assessment phase often stored in unlabelled food containers (34, 59) or in
where physical and chemical hazards are evaluated and baby’s cots (80), with children (particularly older chil-
the contents of the laboratory are determined; and the dren) often enlisted to assist in manufacture. In one case
processing phase where evidence is collected and chemi- a child described assisting a parent during manufacture
cals are removed (68). Entry during these phases is longer of methamphetamine where fumes were present and only
than the initial seizure phase and while PPE may be the adult was using a respirator. These types of exposure
used during these exposures (at different levels depend- are chaotic and not controlled, and differ significantly
ing on the risk) there is limited information on long-term from the type of exposure that occurs with the medical use
health effects associated with repeated investigations/ of ADHD drugs or even drug use (not smoking).
exposures. As with first-responders there are no pub-
lished data on biological monitoring that may be under-
taken to evaluate exposures by long-term investigators to
Neighbours
methamphetamine.
In the US, most clandestine methamphetamine labora-
tory incidents occurred in residential areas, with a quarter
Children
reporting injuries, of which a third are reported to be to
the general (unspecified) public (81). In Australia, 71% of
Children are more sensitive and considered to be at higher laboratories detected were in urban residential areas (18,
risk than adults who may also be present within a clan- 82).
destine drug laboratory as their physiological (early life
Based on US data from 2000 to 2004 (83), approxi-
developmental processes) and behavioural characteris- mately 13% of methamphetamine events (reported as
tics [crawling, mouthing of hands and objects, floor play emergencies) required evacuation of people from neigh-
(59)] result in a higher level of contact with contaminated bouring premises (with 1–300 people evacuated) for a
surfaces (34, 69–73). Children have higher metabolic and median of 3 h. Vapours emitted from ventilation exhaust
respiratory rates (69, 71) and the developing CNS is more fans are at high enough concentrations to corrode metal
sensitive than adults when exposed to some chemicals. fittings (72), and these vapours are commonly discharged
Gastrointestinal absorption differs and the development from premises directly towards neighbours. Waste chemi-
of the skeletal system results in the accumulation of some cals dumped in wastewater, drains, roadside waste and
metals (34).
in public areas comprise corrosive, toxic and flammable
Unauthenticated
Download Date | 10/11/16 7:20 AM
336 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
chemicals and pose a significant hazard to the general vomiting; dizziness; headache; anxiety; bad taste and
public and the environment (62).
lethargy (5, 31, 34, 61, 71, 74, 83, 98). Exposures to high
While information is limited in Australia in relation to concentrations of solvents are associated with liver and
exposures by neighbours, a number of more recent news- kidney effects (5). Accidental ingestion of methampheta-
paper articles have highlighted concerns in relation to mine by children has been associated with (7): agitation
these exposures (82, 84, 85). In addition a number of clan- [most common (99)], tachycardia [second most common
destine laboratories have been detected on the basis of (99)], hypertension, hyperthermia, rhabdomyolysis,
complaints from neighbours in relation to strange odours altered mental status, roving eye movements, cortical
(86, 87).
blindness, ataxia, constant movement, seizure, flailing
No quantitative data is available in relation to the head, neck and extremities, hyperactivity (30), acute res-
levels of contamination that may be present within neigh- piratory symptoms (100) and increased irritability/incon-
bouring premises.
solable crying (73). Children removed from homes used for
the manufacture of methamphetamine are often reported
to smell “like cat urine” as a result of the by-products of
Health effects
methamphetamine production (59, 75, 101, 102).
The most common acute adverse health effects
The available data (34, 61, 70, 73, 75, 88) are considered reported by first responders attending methamphetamine
sufficient to support that a range of individuals, including laboratories include: chemical burns; collapse; abdomi-
children in clandestine drug laboratories are at high risk nal pain; headache; respiratory irritation and effects
for injury and illness associated with immediate hazards (including breathlessness, bronchitis, cough, emphy-
such as fires, explosions and chemical incidents, as well sema, pneumonia and wheezing); skin irritation; central
as acute and chronic exposure to the range of chemi- nervous system effects and mood swings (11, 35, 65, 66, 68,
cals used to manufacture the drugs as well as the drugs 86, 102–105). A volunteer fire-fighter’s lung capacity was
themselves.
found to decrease by 85% after attendance at an explo-
sion at a methamphetamine laboratory (11). The available
studies suggest that 93% of first-responders are likely to
Acute hazards and effects
seek medical treatment for effects and injuries reported
from methamphetamine laboratories (61). No data is avail-
In relation to the operation of clandestine drug laborato- able that provide results of any biological monitoring that
ries, the most significant adverse effects are those derived may have been undertaken to further evaluate the poten-
from immediate acute hazards. These hazards include:
tial for exposure by first-responders.
– The uncontrolled and unprotected storage and use of
volatile, flammable or reactive chemical precursors.
These chemicals may be explosive when mixed.
Chronic effects
– The release of high concentrations of toxic gases
(where these depend on the method of manufacture Chronic health effects of exposure to methamphetamine
but may include ammonia or phosphine) into a room are very poorly understood (71), particularly in relation
or home where ventilation is limited and there is the to environmental exposures to low concentrations, com-
potential for unprotected exposures.
pared with high doses associated with drug use. However,
they may include: neurochemical changes in areas of
Explosions and fires in clandestine drug laboratories the brain that are associated with learning, potentially
have resulted in the death of cooks (33, 42, 60, 89, 90) and affecting cognitive function, behaviour, motor activity
children living in the home (74) or significant chemical, and changes in avoidance responses (106); psychotic,
thermal and inhalation injuries (72, 83, 89, 91–96) that physiological and behavioural/developmental effects
often require higher levels and longer duration of treat- that include violent behaviour, depression, irritability,
ment when compared with other burns injuries (27, 97).
hallucinations, mood swings, paranoia, mood and sleep
Effects consistent with those derived from the range disorders that are associated with exposure to, or use of,
of chemicals and drugs stored and used in the clandestine methamphetamine (75, 106–110); as well as effects asso-
laboratory include: death; burns and irritation of skin, ciated with exposure to the range of chemicals present,
eyes, nose and throat; lacrimation; pulmonary oedema; that includes cancer and effects on respiratory, renal,
coughing; chest pain; shortness of breath; nausea/ hepatic, neurological, developmental and reproductive
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
337
systems (5). Exposures by first-responders have resulted deficits. However, these effects (as well as other cognitive
in chronic respiratory (including asthma and significantly functions) were not apparent at 3 years of age.
decreased lung function), gastrointestinal, neurological
A neuroimaging study of 26 methamphetamine
and immune system effects (29, 63, 102, 111).
exposed (prenatal) and non-methamphetamine exposed
Children removed from homes where methampheta- children (128) suggested an abnormality in energy metab-
mine has been manufactured (112–116) have been reported olism (increased creatine in the striatum) in the brains of
to display a range of behavioural issues including aca- children prenatally exposed to methamphetamine. These
demic difficulties (12), developmental delay (78), a higher changes were not found to be associated with any increase
incidence and risk of externalising (acting out) problems in reported behavioural changes in the children. Further
(112–116), aggressive behaviour (112–116), post-traumatic studies have identified that methamphetamine expo-
or dissociative symptoms (114, 115) and internalising prob- sure during brain development affects the hippocampus
lems (115). In addition children in environments where (responsible for higher cognitive functions) (129) and
methamphetamine, and other drugs or abuse, are used results in cognitive impairments (130) and delayed long-
or manufactured can also be exposed to a wider range lasting memory deficits (131) in adolescent mice.
of other chemicals, neglect, criminal behaviour, abuse
(emotional, physical and sexual) that place these children
at risk of developmental, behavioural and other mental
Confounding factors for evaluating chronic
health problems (114, 115, 117–120).
effects of exposure
It is not clear whether early developmental/behav-
ioural issues of methamphetamine exposure observed Numerous papers (4, 30, 71, 77, 114, 116, 117, 132–136) high-
in children resolve over time, or lead to long-term devel- light issues associated with child welfare, drug use and
opmental problems and a predisposition for addictive methamphetamine manufacturing. Children from homes
behaviours (including drug abuse) later in life (73). Pre- where there is drug abuse and manufacturing frequently
natal exposures (i.e. drug use) to methamphetamine have live in squalor, neglect and abuse (69, 71, 73, 135, 136)
been associated with behavioural problems in children where lack of stimulation, poor nutrition, unsanitary
(increased emotional reactivity, anxiety/depression, conditions and medical problems associated pre- and
externalising and attention-deficit/hyperactivity disor- post-natal exposure to drugs and chemicals (12, 69). Chil-
ders) in children aged 3 and 5 years (121) suggesting the dren from homes with a history of parental drug abuse
potential for long-term development effects. There are few or from a home with domestic violence were 3–3.5 times
studies available, however, where follow-up data has been more likely to test positive to illicit drugs in urine or hair
collected. The most extensive study involved a study on (137). When evaluated, children in methamphetamine
prenatally exposed children from birth to 14 years of age homes showed higher levels of aggression than others
in Sweden (122–126). While there are limitations with the where it is suggested that there is the need to assess the
study (small size of 65 children and no control group) at mental health of children removed from methampheta-
4 years of age the study suggested that the children exhib- mine homes (112, 116). It is suggested that the combina-
ited aggressive behaviour that seemed to correlate with tion/accumulation of multiple risk factors have a greater
longer in-utero exposure periods. The study identified negative impact on psychological development (71) than
that parental drug and alcohol use (prenatal and while the the individual factors alone.
children are growing up), along with other family factors
The U.S. Drug Endangered Children Program that was
influence children’s growth and development. The study created by the San Diego District Attorney’s Office as a solu-
does not specifically correlate only prenatal methamphet- tion to the increasing problem of children removed from
amine exposure with long-term developmental or behav- their parents as a result of the parents arrest for metham-
ioural effects as these are confounded with a wide range phetamine production (74). The multi-agency programme
of other factors associated with parental abuse of drugs that includes procedures/protocols for the decontamina-
and alcohol, criminality, mental health issues, poverty tion and medical assessment of children removed from
and family living arrangements.
these homes, and issues associated with the removal of
A study of potential developmental effects (motor children from these homes has been adopted in some form
skill and cognitive function) of prenatal exposure on 166 by a number of US states (30, 70, 75–77). Europe has estab-
children aged 1, 2 and 3 years (74 exposed and 92 in the lished the European Network for Children Affected by Risky
control group) (127) found that at 1 year of age the meth- Environments within the Family (ENCARE), however, this
amphetamine exposed children had fine-motor skill programme focuses more on children living with parental
Unauthenticated
Download Date | 10/11/16 7:20 AM
338 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
alcohol misuse or domestic abuse. No such programmes (b) absorption of chemicals via the skin; (c) how much
are known to be present in any Australian state.
surface residue sticks to the skin and other objects and
can then be swallowed when placed in the mouth; and
(d) once ingested, how much is absorbed by the body.
Quantification of exposure
While evaluations are available that generally address key
factors that influence exposures by children to environ-
mental contaminants (153), there are a data gaps in this
General
information and more specifically in the data directly rel-
evant to exposures to methamphetamine contamination
The most common approach adopted for the quanti- derived from former clandestine laboratories. These data
fication of exposures by children, and others, to the gaps include (153) methods for monitoring and measuring
presence of methamphetamine and other chemicals children’s exposures and activities, collection of activity
associated with the manufacture of methamphetamine pattern data for children (relevant to all routes of expo-
is to measure concentrations in media relevant to expo- sure), collection and use of data on environmental con-
sure such as indoor air and surface residues. Chemical taminant concentrations on all media of concern [that
intakes of these chemicals are then estimated on the may need to include carpets and soft furnishings (151)],
basis of the measured concentrations and parameters whether exposures associated with indoor air levels of
that estimate physiological characteristic (such as body methamphetamine of importance, dermal transfer coef-
weight), behavioural patterns (such as the time spent ficients and the long-term persistence of surface residues.
in contact with contaminated surfaces) and absorption. In addition data are lacking on the level of exposure that
This approach is consistent with national risk assessment may occur in a former drug laboratory where no remedia-
guidance in Australia (138). The approach is adopted in tion has occurred.
Australia (3, 13), New Zealand (139) and many states in
Some of these data gaps have been addressed using
the US (49, 140–151) for the derivation of assessment and assumptions or estimates in the development of Austral-
remediation criteria for methamphetamine laboratories. ian and international guidelines by using information
These guidelines have been established to be protective obtained on the behaviour and potential for exposure to
of exposures to children, the most sensitive individuals pesticides inside homes (49, 151). The relevance of these
who may be exposed to contamination.
assumptions is not known, particularly where the nature
It is noted that the development of a remediation cri- and behaviour chemical contamination from the opera-
teria for methamphetamine on surfaces inside a home is tion of a clandestine laboratory is likely to differ from
based on a post-remediation exposure scenario (49). This known pesticide applications.
scenario assumes that some remediation of a property has
More recent studies are available defining potential
occurred that removes dusts and other contaminations exposures from indoor air, dermal contact, transfer effi-
that could become re-suspended in the air, and that “res- ciencies and absorption (46, 54, 154–156). These data
ervoirs” of methamphetamine contamination (such as suggest:
contaminated air conditioning filters and ducts and fans)
– There is the potential for methamphetamine in indoor
are not present (49). As a result the key pathways of expo-
air to accumulate in skin oil, clothing, bedding,
sure addressed in the development of the guidelines relate
upholstery and fabric adding to potential oral intakes
to dermal contact with surfaces and objects (accounting
by young children mouthing these types of items
for approximately 80%–95% of total intake) and ingestion
(156). In addition there is the potential for significant
of contamination from mouthing hand and objects (3, 49).
dermal absorption (155). Indoor air pathways have
It is also assumed that since remediation has been under-
not been considered in the development of existing
taken, the remaining contamination degrades on indoor
guidelines.
surfaces and depletes over time with cleaning such that
– The proportion of methamphetamine that may be
exposures are considered to be sub-chronic (occurring
transferred from surfaces to skin is higher than
for < 10% of a lifetime) (152). Exposures in former drug
assumed in the development of existing guidelines
laboratories were not considered to be chronic.
(46, 54, 154).
To quantify chemical intakes from exposures within
a former methamphetamine laboratory requires having The approaches commonly used to evaluate exposure
enough information and data to define (a) where and involve the characterisation of contamination in the envi-
how children may contact these chemicals in the home; ronment where exposure may occur (i.e. measure the
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
339
exposure concentration on/in different media) and/or use
sensitive for the assessment of environmental expo-
biological data to evaluate how much contamination has
sures by more sensitive individuals such as children.
been taken into the body during exposure.
– Few of the available studies relate to samples col-
lected from actual seized laboratories (36, 47, 157).
The majority of the data is from controlled cooks that
Measurement of exposure concentrations
are associated with the manufacture of small quanti-
ties of methamphetamine [noted to be approximately
No data are published or available from other sources in
3 g (44)]. There are no data that enable an assessment
relation to levels of contamination within clandestine
of the relationship of quantitative measures from the
laboratories in Australia. Most of the published data are
controlled cooks to those that may be derived from
available from the US, specifically a number of studies
actual laboratories where larger quantities of meth-
conducted by the National Jewish Medical and Research
amphetamine are produced.
Center. These studies have provided measurements of
– There are no specific data that cover a range of hous-
contamination levels from seized laboratories (noted to
ing types (including different layouts and ventila-
be a limited data set collected after the laboratories were
tion), consideration of different actions/activities that
seized, not operational) and from “controlled cooks”.
may be undertaken by the cooks during manufacture
The controlled cooks enabled the measurement of
(that may change the generation and distribution of
methamphetamine in air and on a range of surfaces (hard,
contamination in a property), and consideration of
soft and clothes) within the cook area and in other areas of
different qualities manufactured.
the premises away from the cook area, as well as volatile
– A limited number of test subjects were evaluated for
organic compounds (VOCs), acids, iodine and phosphine
measurement of residues on individuals (personal
in air. These studies are relevant to a range of metham-
samples) conducting a range of indoor activities fol-
phetamine cook methods and generally address three
lowing the controlled cook of methamphetamine (56).
phases of the operation – cooking of methamphetamine
This limits the overall conclusions that can be drawn
(prior to salting out phase), salting out of methampheta-
from the data presented.
mine and at the completion of the cook.
– No data are available in relation to the potential for
A summary of the data from the available published
systemic absorption of methamphetamine (charac-
studies is presented in Tables 1–3. These relate to the pres-
terised by biomonitoring data) by anyone involved
ence of methamphetamine, and some other chemicals
in the cooking of the drugs, seizure of the laboratory
associated with the manufacture of methamphetamine, in
and subsequent investigation of any of the premises
air and on a range of surfaces from controlled or simulated
evaluated or from exposures that may occur in the
cooks where some data relate to simulated activities in the
premises should no remediation occur.
premises following a cook. It is noted that that level of con-
tamination reported is dependent on the cook method and Exposures in clandestine laboratories are not just limited
the volume of drugs produced. The higher concentrations to the manufactured drug itself. Most of the available data
have typically been reported in actual laboratories where relates to the presence of methamphetamine in the envi-
there has been an explosion. Hence there is a wide range ronment, with some studies also reporting precursors and
of levels of contamination reported from these studies.
by-products that include ephedrine, pseudoephedrine,
None of the published studies provide any data on iodine, hydrogen chloride gas, ammonia gas, phosphine
health effects experienced or biological data from any of gas, total volatile organic compounds and amphetamine.
the individuals exposed.
None of the studies provide analysis of all precursors,
Assessment of aerosol sizes generated during con- intermediates, wastes and products of the manufacture
trolled cooks (44) indicates that most of the methamphet- of methamphetamine that contribute to the mix of chemi-
amine aerosols present in air after a cook are respirable, cals to which anyone within the laboratory, including chil-
with up to 90% < 1 μm in diameter.
dren may be exposed (158). Reviews of the wide range of
A number of limitations have been identified in rela- chemicals that may be associated with the manufacture
tion to the available data, in particular:
of methamphetamine (3, 159), on the basis of the nature,
– The majority of the studies conducted by the National behaviour (including persistence) and availability of data
Jewish Medical and Research Center (36, 38, 39, 47, that can be used to characterise exposure, identified
48, 50, 56) used occupational-exposure based analyti- a number of key chemicals that can be used as reliable
cal methods. These methods may not be adequately indicators for the manufacture and exposure to chemicals
Unauthenticated
Download Date | 10/11/16 7:20 AM
340 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
Table 1: Summary of methamphetamine and other chemicals in indoor air.
Location/activity
Range of maximum concentrations reported in air (μ
g/m3)
References
MA
Hydrogen Phosphine
Ammonia Iodine
chloride
Data from seized laboratories (cook methods not specified)
Range of different rooms from 0.17–7.3
190–200
nd to 358.6
– 10–23
(36, 47, 51)
seized laboratories – after the
cook
Suspected clandestine drug
0.2–3
–
–
– –
(58)
laboratories (9 locations)
Data from controlled cooks – anhydrous ammonia method
Within cook area
– Cook phase
10.1–34
–
–
– –
(38)
– Salting out
127–680
–
–
– –
– Post cook
7.6–79
895–1044 –
90,500–286,000 –
Away from cook area
– Cook phase
2.4–42
–
–
– –
(38)
– Salting out
12–158
–
–
– –
– Post cook
7.6
596
–
< 46,000–255,000 –
Data from controlled cooks – red phosphorous and hypophosphorous methods
Within cook area
– Cook phase
< 0.19
119–313
–
– nd to 29 (36, 39, 44, 47, 50)
– Salting out
680–5500
220–30,000 –
– nd to 25
– Post cook
79–5500
75–14,600 nd to 18,000
– 52–1600
Away from cook area
– Cook phase
< 0.17
30
–
– nd to 5
(44, 47, 50)
– Salting out
960–4000
390–6710 –
– –
– Post cook
2.6–4200
30–313
–
– 5–156
Day following cook for no activity, 70 (no activity) nd to 67
–
– nd to 26
(44, 50)
medium and high activity (up to
–210 (high
18 hrs post cook) (1 cook) (red
activity)
phosphorous method)
MA, Methamphetamine; nd, not detected (variable analytical limits or reporting); –, no data reported for analyte.
from methamphetamine laboratories. These key chemi- not be used as a surrogate if the laboratory were only used
cals include those commonly reported in the available for the manufacture or extraction of pseudoephedrine.
studies.
A laboratory study (160) in relation to the recovery of
pseudoephedrine and methamphetamine residues from
Sampling and analysis issues
impermeable surfaces (glass, stainless steel, adhesive
vinyl laminate, stone benchtop, varnished floor wood, A range of analytical methods have been used in the
painted metal sheet and varnished benchtop wood) sug- measurement of contamination (on surfaces and in dif-
gested that methamphetamine can be used as a surrogate ferent materials) associated with clandestine laboratories
to represent both methamphetamine and pseudoephed- (157, 160–166).
rine (where methamphetamine has been synthesised) on
For the measurement of contamination on surfaces in
impermeable surfaces from clandestine drug laborato- premises, wipe sampling methods are commonly used. A
ries. It is noted that data from actual seized laboratories study of the efficacy of wipe sampling methods (167) iden-
(36) suggests this is reasonable for most surfaces with the tified that it was appropriate to use either methanol or iso-
exception of appliances within kitchens (such as micro- propanol wipes for the collection of the samples and that
wave ovens) that are used in the manufacture of drugs the presence of dust or paint on the wipe samples did not
where the proportion of pseudoephedrine (precursor more interfere with the analytical results. The recovery of meth-
likely to be used in these appliances) has been found to be amphetamine from surfaces using wipe sampling is vari-
higher than methamphetamine. Methamphetamine could able depending on the nature of the surface. Recoveries
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
341
Table 2: Summary of amphetamine and precursor residue levels on hard surfaces.
Location/activity
Range of maximum contaminant surface residues reported
References
(μ
g/100 cm2)
MA
AMP
EPH
PSE
Data from seized and suspected laboratories (cook methods not specified)
Wal s and surfaces that include benches, tables, 0.1–6093 to 16,000 1.2–34 6.6–120 99–1400
(36, 47, 51, 157)
floors, fans, appliances
after explosion
Ventilation fans
0.2–450
nd to1.2 nd to 6.6 0.5–99
(36)
Kitchen appliances (microwaves, burners, ovens, nd to 16,000
nd to 33 nd to 1200 nd to 51,000
(36)
refrigerators
After 3 rounds of decontamination
0.14–1.05
–
–
–
(158)
Data from controlled cooks–anhydrous ammonia method
Various surfaces (3 cooks)
0.08–160
–
–
–
(38), (47)
Data from controlled cooks–red phosphorous method
Various surfaces (2 cooks)
6.1–68*
–
–
–
(44, 50)
Data from controlled cooks–hypophosphorous method
Various surfaces (painted wal , glass, mirror) up to 0.078–23
–
–
–
(39)
7 feet from cook area (2 cooks)
Various, including within hotel room
0.1–860
nd to 3.2 nd to 0.5 nd to 2.6
(36, 47)
MA, Methamphetamine; AMP, amphetamine; EPH, ephedrine; PSE, pseudoephedrine; nd, not detected (variable analytical limits or
reporting); –, no data reported for analyte; *, surface residue levels similar immediately post cook, 13 h post cook, 16 h post cook and 18 h
post cook.
of methamphetamine residues from surfaces have been (170). In general amphetamines are weak bases with low
reported to be < 100% (51, 167), with specific studies indi- protein binding (173) and have a high volume of distribu-
cating variability between 15% for porous surfaces and tion which means almost all of the total amount of drug
80% for smoother surfaces (160).
available in plasma may diffuse across cell membranes
In relation to the analysis of methamphetamine, the and lipid layers to tissue matrices with lower pH values
available studies suggest the variability between laborato- than blood (174). Saliva/oral fluid, sweat and breast milk
ries ranges from 3%–30% (167) to 1%–50% (51).
are more acidic than plasma, hence amphetamines are
These studies indicate that sampling and analysis readily distributed to these fluids (174, 175).
methods can detect the presence methamphetamine,
Extensive reviews of the metabolism of methampheta-
with the level of recovery varying between porous and mine and amphetamine are available in the literature (170,
smooth surfaces. In addition some variability in the levels 176). These mechanisms do not appear to be changed by
reported by different laboratories (between 1% and 50%) chronic exposure (177). The major pathways of metham-
can occur. This should be considered where quantitative phetamine metabolism involve (170, 176, 177):
data from different surfaces and laboratories is compared.
– n-demethylation to form amphetamine, that can then
be metabolised via several pathways
– aromatic hydroxylation to form 4-hydroxymetham-
Measurement of exposure using biological
phetamine and then 4-hydroxyamphetamine and
data (Biomarkers)
4-hydrocynorephedrine
– β-hydroxylation to form norephedrine.
General
There are a number of metabolites that are produced from
Amphetamines are readily absorbed via inhalation [with these mechanisms, with amphetamine and 4-hydroxy-
between 67% and 79% (168) and 90% (169) absorbed into methamphetamine being the major metabolites detected
the blood stream], ingestion [with oral bioavailability in urine. In addition amphetamine is a major drug of
noted to be in the range of 67.2% (170, 171) to 85% (172)] and abuse, and it may also be present as an impurity or
dermal pathways (45). Following intake, amphetamines mixture with methamphetamine. Evaluating the pres-
are rapidly distributed to the major organ systems includ- ence and ratios of methamphetamine and amphetamine,
ing the brain as it readily crosses the blood-brain barrier both of which have relatively long elimination half-lives
Unauthenticated
Download Date | 10/11/16 7:20 AM
342 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
Table 3: Summary of amphetamine and precursor residue levels on individuals, clothes, soft furnishings and toys.
Location/activity
Range of maximum contaminant residues
References
reported (μ
g/sample, many as μ
g/100 cm2)
MA
AMP
EPH
PSE
Data from seized laboratories (cook methods not specified)
Window furnishings and sofa
0.84–120
nd to 1
nd
0.9–12
(36)
Carpet
132–2045
–
–
–
(51)
Data from controlled cooks–red phosphorous, hypophosphorous and anhydrous methods
Personal samples from cooks (2–7 cooks)
– Cook phase
nd to 19.3
–
–
–
(36, 38, 39, 47, 56)
– Salting out
nd to 580
–
–
–
– Post cook
0.2–150
–
–
–
Personal samples from investigators (5 cooks)
– Cook phase
nd to 0.14
–
–
–
(56)
– Salting out
2.54–580
–
–
–
– Post cook
1.1–150
–
–
–
Personal samples – post cook (5 cooks)
– Police
nd to 1.6
–
–
–
(56)
– Fire fighter
0.46–56
–
–
–
– Juvenile
nd to 1.18
–
–
–
– Child (simulated crawling by adult)
0.2–29
–
–
–
Personal wipe samples –post cook
– Low activity
0.075–1.7
–
–
–
(44, 50)
– Medium activity
0.32–56
–
–
–
– High activity
0.59–44
–
–
–
Personal samples after decontamination (2–7 cooks)
0.43–10.2
–
–
–
(38, 39, 56)
Dog (5 cooks)
1.89
–
–
–
(56)
Baby clothes near cook (2 cooks)
6.4–500
–
–
–
(39)
Toys (including teddy bear)
6.4–1300
–
–
–
(36, 39)
Carpet
3.93–13
–
–
–
(36)
Carpet – vacuum samples (μg per m2)
54–270
–
–
–
(44, 50)
MA, Methamphetamine; AMP, amphetamine; EPH, ephedrine; PSE, pseudoephedrine; nd, not detected (variable analytical limits or
reporting); –, no data reported for analyte.
in the body making them detectable in various biological Excretion can be increased by urinary acidification, and
matrices, provides an indication of systemic absorption decreased by urinary alkalinisation (170, 174).
of methamphetamine and/or amphetamine. Follow-
Due to the rapid absorption and excretion of metham-
ing intake of pure methamphetamine, the presence of phetamine and metabolites the detection times for meth-
amphetamine relates to the metabolism of the primary amphetamine in most biological matrices are short. The
drug and the ratio of methamphetamine to amphetamine detection times differ depending on whether exposure
should be > 1 (178). Hence the presence of both metham- occurred from a single dose, repeated doses or chronic
phetamine and amphetamine in biological matrices are exposures. Most data are available following a single dose
commonly used as indicators of systemic absorption of where the detection time is reported to range from 24 to
methamphetamine.
48 h in plasma to 87 h in urine (177). Limited data are
Methamphetamine, amphetamine and their metab- available in relation to repeated doses of methampheta-
olites are excreted primarily in urine, with 55%–69% mine, however, the detection time is in the range of 3 days
excreted in the first 24-h after exposure (170). Based on in saliva/oral fluid to 8 days in urine and sweat (177, 179–
studies associated with doses typically associated with 181). Accumulation of amphetamines in a keratin matrix
drug use, an average of 30%–40% of a methamphetamine is more complex (174) but has been shown to provide a
dose is excreted unchanged and the remainder is elimi- stable measure of temporal exposures with the distribu-
nated as metabolites (170). As amphetamines are weak tion of drugs along the shaft of the hair expected to reflect
bases, renal excretion is variable and is dependent on pH. historical month-by-month exposures (174).
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
343
In relation to the potential for biomarkers to be used the detection limit and allows for low levels to be present
as a reliable measure of environmental exposure to meth- either directly or as metabolites from prescribed medi-
amphetamine (and amphetamine that may be present as cines (182, 184). Methamphetamine and amphetamine
an impurity or as a major metabolite of methampheta- concentrations in urine are generally higher than reported
mine), review of these biological matrices has considered in blood plasma and, while rapidly cleared from the body,
the following factors that are considered to be important can remain quantifiable for longer periods of time after
for utilising the data in a study that relates to evaluating multiple doses, with detections reported after 46–196 h
potential environmental exposures:
(181).
1. The potential for the biomarker to be present in the
The testing for methamphetamine and amphetamine
matrix sampled, and be a stable measure of exposure; in urine is often conducted upon hospital admission to
2. The potential for the biomarker to report positive evaluate drug use. Methamphetamine cooks treated in
detections, if exposure occurred, at the point in time hospital for various injuries associated with drug manu-
when samples can be collected (may be longer than facture commonly (around 91%) test positive for ampheta-
a week);
mines (29, 89).
3. The potential for data to be easily collected; and
One study is available where urine samples have been
4. The potential for the analysis to be able to report detec- collected from 104 children removed from methampheta-
tions, if exposure occurred, that relate to environmen- mine laboratories (37). The children were tested at emer-
tal exposures from the clandestine drug laboratory.
gency medical departments immediately after removal
from the premises where 46% of the children reported
These aspects have been considered further in relation to positive detections (reported as detections only, no quan-
the use of blood and urine, saliva/oral fluid, sweat and titative data) for methamphetamine. Of the children who
hair for the potential assessment of environmental expo- tested positive, 85% were 8 years old and younger. No
sures. The use of these matrices for the assessment of child tested positive more than 6.5 h after removal from
exposure to amphetamines in the literature has primarily the laboratory highlighting the importance of the ability
focused on users, with limited data available for environ- to collect urine samples within the window of detection.
mental exposures. Where data is available that relates to No information or data is available from this study on the
environmental exposures much of it is presented as a pos- levels of methamphetamine (and precursors) within the
itive or negative finding, rather than a quantitative value. homes from which the children were removed.
Given the rapid clearance of methamphetamine and
metabolites from the body, blood plasma or urine are not
Blood and urine
considered to be a suitable indicator of former environ-
mental exposures, where sample collection may only be
Blood plasma is the most direct quantitative measure of possible more than a week (and likely longer) following
the level of methamphetamine and amphetamine within the cessation of exposure.
the body at a point in time following exposure. The half-
life of methamphetamine in plasma varies from 9.1 to
13.1 h with a window of detection for the presence of the
Saliva/oral fluid and sweat
drug in plasma up to 24 h (181) following exposure. In
plasma, after oral administration of methamphetamine, Saliva/oral fluid has been identified as an easily accessi-
concentrations of the metabolite amphetamine are lower ble and suitable biomonitoring method for the assessment
than methamphetamine with the 24-h area under the of drugs of abuse (179). A number of studies have indi-
curve (AUC ) for amphetamine showing a typical dose- cated that oral fluid methamphetamine concentrations
24
response relationship (169, 171, 181).
are higher than blood plasma (169, 171, 179, 181), however,
As urine is the primary mechanism of elimination fol- there was a poor correlation between saliva/oral fluid and
lowing exposure to amphetamines, it is most commonly plasma methamphetamine concentrations reflecting high
used for the purpose of assessing and quantifying work- intra and inter-individual variability. While some attempts
place exposure, driving related offences and criminal have been made to better define saliva-plasma ratios (S/P)
cases (181–183). Analyses of urine for exposure to meth- for methamphetamine (171, 185) the measure is generally
amphetamine are only considered positive if the levels are not considered to be a reliable quantitative measure of
above a pre-determined cut-off limit and the metabolite exposure, and is only considered to be a suitable matrix
amphetamine is also detected. The cut-off limit is above for screening for drug use (181).
Unauthenticated
Download Date | 10/11/16 7:20 AM
 344
344 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
The testing of sweat using sweat patches is a non- activity and cell division associated with the anagen (i.e.
invasive method of biomonitoring, however, only a formation of the hair shaft) growing phase of the hair
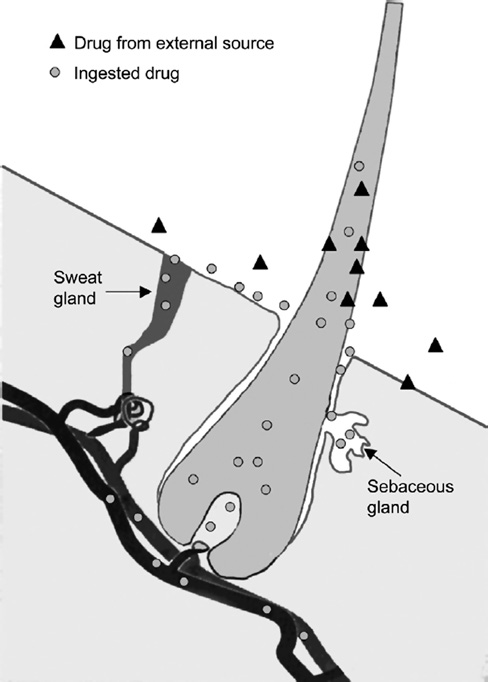
limited number of studies are available that assist in the (190). There are three recognised routes by which drugs
understanding of methamphetamine and amphetamine are incorporated into the hair, as illustrated in Figure 4.
excretion in sweat (180, 186). Testing conducted with These include incorporation of drugs from the circulatory
other drugs has identified some uncertainties associated system (191); absorption from sebum and sweat bathing
with the method that include potential for time-depend- the hair; and from external contamination (190).
ant drug loss due to drug degradation, reabsorption to
Within the hair itself, the drugs and metabolites are
the skin, volatile losses and contamination on the skin incorporated/bound into the keratinaceous matrix of the
(180, 187). In relation to methamphetamine and ampheta- hair shaft during protein synthesis. In the hair shaft, the
mines, the available studies indicate that sweat testing materials form a stable drug bolus that remains embedded
is an effective and reliable test for detecting drug use, in the hair matrix. Different drugs have different affinities
however, significant intra- and inter-individual variability and binding capabilities which vary depending on drug
indicated it should only be used as a qualitative screening pKa, structure, size, lipophilicity, protein binding capac-
test to report positive detections rather than a quantitative ity and melanin affinity (190). The lipid solubility of a
test (180, 186).
drug is a critical factor for the transport of the drug from
Given the rapid clearance of methamphetamine and the blood stream across the cell membrane and into the
metabolites from the body, and the variability issues iden- growing hair (190).
tified in relation to the use of saliva/oral fluid and sweat,
In sufficiently long hair, sectional analysis can
these media are not considered to be a reliable quantitative provide a timeline of drug exposure/use (191, 192). The
method for the assessment of environmental exposures.
drug is incorporated into the hair matrix as it grows with
the growth rate approximately 2.8–3.2 mm per week (an
Nails
Few studies are available that specifically address the use
of nails as an analytical media for the detection of drugs
(188). The available studies indicate that fingernail and
toenail clippings have been found as reliable as hair for
the detection of methamphetamine and amphetamine
in users, as these drugs are well accumulated in the nail
matrix, stable in the nail, retained for a long period of
time, show a good correlation with hair concentrations
(174, 188, 189). The mechanism of deposition at the nail
matrix is complex (188, 189), hence analysis of nails are
considered to be a less reliable indicator of temporal
trends than hair. However, analysis of nails may provide
an alternate method of evaluating environmental expo-
sures to methamphetamine.
Hair
General
The incorporation of drugs and metabolites into hair has
been found to provide a reliable basis for evaluating his-
torical use or exposure (190). The mechanisms by which
drugs and their metabolites are incorporated into hair are
complex and not fully understood (190). Conceptually it
is believed that drugs and their metabolites (as well as
other trace elements) are incorporated during metabolic
Figure 4: Routes of drug incorporation into the hair follicle (190).
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
345
average of 1 cm/month) and clearance of the drugs from methamphetamine, the ratio of amphetamine to meth-
the follicle cells during the 5–8 days after exposure (174). amphetamine in hair is reported typically to be approxi-
The testing of drugs in hair has a long window of detection mately 1:10 (174), however, it is noted that this ratio has
and the samples can easily be collected and stored under been found to increase with the duration of drug abuse
a range of normal conditions (193).
(192) and presumably environmental exposures.
The window of detection is limited by the length of the
Melanin has been proposed as an important factor in
hair (relevant to systemic absorption where the window the incorporation of amphetamines in hair (174, 197, 198).
of detection can range from weeks to months) and, where While the nature of the interaction has not been estab-
environmental exposures are concerned, the cleanliness lished a significant correlation has been observed in con-
of the hair (deposition onto hair) (193).
trolled human studies (199).
Factors that can affect the stability of drugs in hair
relate to the morphology and physicochemical properties
of the hair as well as external factors such as exposure to
Dose response
sunlight and weathering, dying, bleaching or treatment of In general, hair analysis can be used to approximate
hair and curling or straightening (which damages the hair dose. The mechanism of entrapment suggests that there
shaft) (190).
should be a pharmacological relationship between the
intake of a drug and the amount of drug or metabolite
Incorporation of amphetamines in hair
recovered from hair (191). A positive linear relationship
Hair testing is considered to be a reliable biological and between dose and hair concentration has been identified
stable marker for cumulative and temporal measure of for cocaine and medicinal drug use (200) with segmented
exposure to amphetamines, with a long window of detec- analysis of hair used to evaluate changes in dose over
tion making it suitable for the assessment of exposure time (201, 202). In relation to use of methamphetamine,
even after a long period of time has elapsed since expo- a positive dose-response relationship has been demon-
sure occurred.
strated with rat hair (203), in drug users (204) and in a
The first study in relation to the incorporation of controlled study (199).
methamphetamine in hair was in 1954 in a guinea pig,
The relationship from these studies, however, may
with a large number of animal studies further conducted not be used to determine dose from the hair analysis
to evaluate the incorporation of amphetamines into the alone as a number of researchers have reported substan-
keratin matrix to investigate the pharmacokinetics (174).
tial inter-individual variability in hair concentrations
Amphetamines absorbed into the keratinaceous (191). It is suggested (191) that some of these variability
matrix have been found to be tightly bound and are stable issues may be due to the variety of assay protocols uti-
over long periods of time (191, 192). Amphetamines, and lised in these studies or melanin concentrations in hair
other contaminants that are externally deposited or not (where a significant correlation has also been observed)
tightly bound can be removed through a series of ethanol (199). Regardless of the variability observed it still holds
or isopropyl alcohol washes followed by phosphate buffer that the higher the dose the higher the concentration in
washes (192). By analysing the concentrations recovered hair. Hence where a single competently executed assay
from the washes to the concentrations recovered from protocol is used it has been found to provide a useful tool
the hair matrix, a determination can be made that distin- in rank-ordering doses (191).
guishes passive or environmental exposures/contamina-
tion from systemic absorption (191, 194). Deposition of
Published data on use of hair analysis to assess
amphetamines from air, such as from smoking or from the
environmental exposures for children
suspension of amphetamine residues in a home during Hair analysis for drugs has been used in a small number
vacuuming or from the operation of a contaminated air of cases of suspected child abuse where proof of harm was
conditioning unit, could be a potential route of entry into required to be demonstrated (205).
hair (195).
Published reports on the use of hair analysis for evalu-
More specifically in relation to methamphetamine ating environmental exposures (i.e. not drug use) to meth-
exposures, analysis of both methamphetamine (from amphetamine in children are limited (98, 193, 205–207).
systemic absorption and deposition) and amphetamine The available data have provided evidence of exposure by
(metabolite following systemic absorption only) has children as summarised below:
been used as a quantitative method of differentiating
– In general, approximately 35%–73% of biological
between the types of exposure (196). From the intake of
samples, as urine and/or hair samples collected from
Unauthenticated
Download Date | 10/11/16 7:20 AM
346 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
children exposed to methamphetamine in the home an operational or former clandestine laboratory have no
(from drug use or manufacture), reporting positive choice (12) in relation to drug exposures and their intake
detections results for methamphetamine, ampheta- and health is not monitored and managed.
mine, pseudoephedrine and/or ephedrine exposures
(30, 37, 70, 71, 78, 88, 133, 207).
– More specifically, between 45% and 73% of children
Analysis methods
[with 100% from one small study of four children In relation to the quantification methamphetamine and
(208)] exposed to methamphetamine via drug use or amphetamine in hair samples, there are a wide range of
manufacture tested positive for methamphetamine methods (192, 193, 196, 206, 209–216) that rely on the sam-
in hair (70, 73, 196, 207). In some cases (where data pling of different quantities of hair (that have the poten-
are reported) positive detections were reported in hair tial to affect the laboratory quantitation limit), potential
where no detections were reported in urine (73).
inclusion of segment analysis (for evaluation of exposure
– Hair analysis of a child injured from the ingestion over time), utilisation of different extraction methods and
of caustic liquid (drain cleaner) in the US (where inclusion of methods for the evaluation of deposited and/
methamphetamine was manufactured in the home) or absorbed contamination. The washing of hair during
reported detections of methamphetamine (1.7 ng/mg) analysis needs to be undertaken with caution as some
and amphetamine (0.16 ng/mg) (98).
methods have the potential to damage the hair shaft and
– Hair analysis data from New Zealand (207) from affect the reporting of absorbed methamphetamine and
children removed from clandestine drug laborato- amphetamine (194).
ries reported 73% detection of methamphetamine in
Where an analytical method is required for the quan-
hair above 0.1 ng/mg and low level detection (10%) tification of methamphetamine and amphetamine in hair,
of methamphetamine determined to be present from it is important that these issues are evaluated and resolved
external contamination/deposition (i.e. in the hair to ensure that data is sufficiently robust.
wash). The levels of methamphetamine reported in
children ranged from 0.1 to 131 ng/mg, with higher
concentrations reported in children under 5 years of
Summary
age.
On the basis of the literature review undertaken it is clear
The actual incidence of positive detections of metham- that the operation of clandestine methamphetamine labo-
phetamines in hair samples, however, may be under ratories results in the presence of a wide range of hazards
reported as many jurisdictions do not conduct medical and risks within the premises including the contamina-
testing on children, or on all children, removed from clan- tion of all indoor surfaces and materials with metham-
destine laboratories and/or do not report these data (due phetamine residues. The operation of these laboratories
to privacy issues) (78).
has the potential to result in significant hazards (primar-
The level of exposure that corresponds with the detec- ily fire, explosion and release of high concentrations of
tion of precursors and drugs in biological samples is not toxic gases) and other acute exposures by individuals
known and is generally poorly understood (4, 12, 34, 37). A who have chosen to conduct the illegal activities (cooks).
study by Weisheit (27) considers that exposures to chemi- However, these activities also has to potential to expose a
cals other than methamphetamine within clandestine range of other individuals, who have not chosen to take
drug laboratories is of greater concern on the basis that on these illegal activities, to the same hazards and risks.
doses of methamphetamine expected to be absorbed by These individuals include children (considered to be the
a child from contaminated surfaces is lower than doses most sensitive group in relation to exposure), neighbours,
received during drug use, and that methamphetamine police and first-responders to a fire or explosion, foren-
is often administered to children with behavioural prob- sic and other local investigators (including local council
lems (such as ADHD). While these arguments suggest a officers) and any residents who may live in these homes
relative understanding of potential exposures, they do before remediation or if no remediation is conducted.
not take into account the voluntary nature of drug use
It is important that these risks are understood such
and monitored/controlled use of ADHD medications. Nor that appropriate measures can be implemented to manage
is the statement based on any evidence of the exposure exposures and/or determine the need for medical evalua-
levels that may occur within a former clandestine drug tion and intervention, particularly if there is the potential
laboratory. Children exposed to methamphetamine in for harm to have occurred. This is particularly relevant
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
347
to children, who are most vulnerable group who have no 14. Law and Justice Legislation Amendment (Serious Drug Offences
choice in where they live, and are exposed to varying levels
and Other Measures) Act 2005 (Cwlth), An Act to amend various
of hazards and chemical contamination in the clandestine
Acts relating to law and justice, and for related purposes, Rule
129 SDO Act.
laboratory. While limited, the available literature provides 15. Misuse of Drugs Amendment Act 2011 (WA).
supporting evidence that shows that children living in 16. WA Health. Interim Guidelines for notification and risk manage-
these homes are exposed to the drugs manufactured, and
ment after detection of a clandestine drug laboratory (Clan Lab).
that these exposures have resulted in adverse acute and
Government of Western Australia, Department of Health, Public
chronic health effects, including long-term behavioural
Health, 2012.
issues. More data are needed to better define these expo- 17. Victoria Health. Clandestine laboratory remediation, Envi-
ronmental health practice note. Melbourne: State of Victoria,
sures, however, the limited data available suggest that
Department of Health, 2012.
further evaluation and the development of appropriate 18. Australian Crime Commission. Illicit drug data report 2009–10.
protocols for the assessment and management of these
Report. Australian Crime Commission, 2011 ISSN 1327–9068.
children needs to be established in Australia.
19. Schloenhardt A. The market for amphetamine-type stimulants
and their precursors in Oceania. Research and Public Policy
Series No. 81. Australian Institute of Criminology, 2007.
20. Wilkins C, Sweetsur P, Smart B, Warne C, Jawalkar S. Recent
References
trends in illegal drugs in New Zealand, 2006–2011, findings
from the 2006, 2007, 2008, 2009, 2010 and 2011 illict drug
monitoring system (IDMS). SHORE and Whariki Research Centre,
1. Australian Crime Commission. Clandestine Laboratories. Crime
Massey University, 2012 July 2012. Report No.
Profile Series. Australian Crime Commission and Australian
21. Newell P. Clandestine drug manufacture in Australia. Chemistry
Government, Attorney-General’s Department, 2011.
Australia 2008;75(3):11–4.
2. Australian Crime Commission. Illicit Drug Data Report
22. Willis K, Homel P, Gray K. Developing and implementing a per-
2013–2014. Australian Crime Commission, 2015.
formance measurement framework for drug law enforcement in
3. Wright J. Derivation of risk-based investigation levels,
Australia. Australian Institute of Criminology, 2006.
clandestine drug laboratory, site investigation guidelines.
23. Australian Crime Commission. Illicit Drug Data Report 2011–12.
Report. Sydney: Environmental Risk Sciences, 2009.
Australian Crime Commission, 2013.
4. Sheridan J, Bennett S, Coggan C, Wheeler C, McMil an K. Injury
24. Australian Crime Commission. Illicit Drug Data Report 2010–11.
associated with methamphetamine use: a review of the litera-
Australian Crime Commission, 2012.
ture. Harm Reduct J 2006;3:14.
25. Australian Crime Commission. Illict Drug Data Report 2012–13.
5. Irvine GD, Chin L. The environmental impact and adverse health
Australian Crime Commission, 2014.
effects of the clandestine manufacture of methamphetamine.
26. Hargreaves G. Clandestine drug labs, chemical time bombs. FBI
NIDA Res Monogr 1991;115:33–46.
Law Enforcement Bulletin, 2000:1–6.
6. Donnermeyer JF, Tunnell K. In our own backyard: methampheta-
27. Weisheit R. Making methamphetamine. J Rural Soc Sci
mine manufacturing, trafficking and abuse in rural america.
2008;23(2):78–107.
Rural Realities [Internet] 2007;2(2):1–12.
28. Vandeveld N. Clandestine methamphetamine labs in Wisconsin.
7. Australian Institute of Criminology. National amphetamine-
J Environ Health 2004;66(7):46–51.
type stimulant strategy, background paper. monograph series.
29. Watanabe-Galloway S, Ryan S, Hansen K, Hul siek B, Muli V,
National Drug Research Institute, Australian Institute of Crimi-
Malone AC. Effects of methamphetamine abuse beyond indi-
nology, 2007.
vidual users. J Psychoactive Drugs 2009;41(3):241–8.
8. Caldicott D, Pigou P, Beattie R, Edwards J. Clandestine drug labo-
30. Grant P. Evaluation of children removed from a clandestine
ratories in Australia and the potential for harm. Aust N Z J Public
methamphetamine laboratory. J Emerg Nurs 2007;33(1):31–41.
Health 2005;29(2):155–62.
31. Burge M, Hunsaker JC, 3rd, Davis GJ. Death of a toddler due to
9. Parliamentary Joint Committee on the Australian Crime Com-
ingestion of sulfuric acid at a clandestine home methampheta-
mission. Inquiry into the manufacture, importation and use of
mine laboratory. Forensic Sci Med Pathol 2009;5(4):298–301.
amphetamines and other synthetic drugs (AOSD) in Australia.
32. Hughart JL. Chemical hazards related to clandestine drug labora-
Canberra: The Parliament of the Commonwealth of Australia,
tories. Arh Hig Rada Toksikol 2000;51(3):305–10.
2007.
33. Scott MS. Clandestine drug labs, problem-oriented guide for
10. Ministerial Council on Drug Strategy. National Amphetamine
police series. 2002.
Type Stimulant Strategy 2008–2011. 2006.
34. Ferguson TJ. Overview of medical toxicology and potential for
11. McFadden D, Kub J, Fitzgetald S. Occupational health hazards
exposures to clandestine drug laboratories in California. Report.
to first responders from clandestine methamphetamine labs.
Minnesota Department of Health, 2003.
J Addict Nurs 2006;17(3):169–73.
35. Gardner G. Illegal drug laboratories: a growing health and
12. Lineberry TW, Bostwick JM. Methamphetamine abuse: a perfect
toxic waste problem. Pace Envtl L Rev 1989;1-1-1989(Paper
storm of complications. Mayo Clin Proc 2006;81(1):77–84.
122):193–212.
13. Australian Crime Commission. Clandestine drug laboratory
36. Martyny JW, Arbuckle SL, McCammon CS, Esswein.EJ, Erb N.
remediation guidelines. Attorney-General’s Department,
Chemical exposures associated with clandestine methampheta-
Commonwealth of Australia, 2011.
mine laboratories. Report. Denver CO, 2004.
Unauthenticated
Download Date | 10/11/16 7:20 AM
348 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
37. Grant P, Bell K, Stewart D, Paulson J, Rogers K. Evidence of
Impact on Meth Lab Cleanup Guidance. Minnestota Pollution
methamphetamine exposure in children removed from clan-
Control Agency, 2007 November 2011. Report No.
destine methamphetamine laboratories. Pediatr Emerg Care
52. Li H. Adsorption and desorption capacity of methamphetamine
2010;26(1):10–4.
in gypsum drywal [Dissertation/Thesis]: Missouri University of
38. Martyny JW, Arbuckle SL, McCammon CS, Erb N. Chemical
Science and Technology; 2014.
exposures associated with clandestine methamphetamine labo-
53. Poppendieck D, Morrison G, Corsi R. Desorption of a metham-
ratories using the anhydrous ammonia method of production.
phetamine surrogate from wal board under remediation condi-
Denver CO: National Jewish Medical and Research Center, 2004.
tions. Atmos Environ 2015;106:477–84.
39. Martyny JW, VanDyke M, McCammon CS, Erb N, Arbuckle SL.
54. Van Dyke M, Martyny JW, Serrano KA. Methamphetamine
Chemical exposures associated with clandestine methampheta-
residue dermal transfer efficiencies from household surfaces.
mine laboratories using the hypophosphorous and phosphorous
J Occup Environ Hyg 2014;11(4):249–58.
flake method of production. Denver CO: Division of Environmen-
55. Serrano KA, Martyny JW, Kofford S, Contreras JR, Van Dyke MV.
tal and Occupational Health Sciences, Sciences DoEaOH, 2005.
Decontamination of clothing and building materials associated
40. Burgess JL. Phosphine exposure from a methamphetamine
with the clandestine production of methamphetamine. J Occup
laboratory investigation. J Toxicol Clin Toxicol 2001;39(2):165–8.
Environ Hyg 2012;9(3):185–97.
41. Bloom GR, Suhail F, Hopkins-Price P, Sood A. Acute anhydrous
56. Martyny JW. Methamphetamine contamination on persons
ammonia injury from accidents during illicit methamphetamine
associated with methamphetamine laboratories. Denver, CO:
production. Burns 2008;34:713–8.
National Jewish Medical and Research Centre, 2008.
42. Willers-Russo LJ. Three fatalities involving phosphine gas,
57. Martyny JW. Methamphetamine stability and recovery on
produced as a result of methamphetamine manufacturing.
painted drywal surfaces. 2008.
J Forensic Sci 1999;44(3):647–52.
58. McKenzie EJ. Chemical contamination in former clandestine
43. McKetin R., McLaren J. The methamphetamine situation in
methamphetamine laboratories. University of Auckland, 2014.
Australia: a review of routine data sources, NDARC Technical
59. Roper JD. Drug-endangered children and the manufacture of
Report No. 172. National Drug Law Enforcement Research Fund,
methamphetamine. School Nurse News 2007;24(2):27–9.
an initiative of the National Drug Strategy, 2004 Contract No.:
60. Cooper D, Hanlon D, Fischer P, Leiker MS, Tsongas T, et al.
Technical Report Number 172.
Public health consequences among first responders to
44. VanDyke M, Erb N, Arbuckle S, Martyny J. A 24-hour study to
emergency events associated with illicit methamphetamine
investigate persistent chemical exposures associated with clan-
laboratories–selected states, 1996–1999. Morb Mortal Wkly Rep
destine methamphetamine laboratories. J Occup Environ Hyg
2000(45):1021–4.
2009;6(2):82–9.
61. Thrasher DL, Von Derau K, Burgess J. Health effects from
45. Hui X, Salocks CB, Sanborn J, Maibach H. In vitro studies
reported exposure to methamphetamine labs: a poison center-
of percutaneous absorption and surface-to-skin transfer of
based study. J Med Toxicol 2009;5(4):200–4.
d-Methamphetamine hydrochloride using human skin, poster at 62. Cameron M. Health and safety concerns for law enforcement
47th Annual Meeting and ToxExpo of the Society of Toxicology.
personnel investigating clandestine drug labs. Chem Health Saf
2009.
2002;9(1):6–9.
46. Salocks CB, Hui X, Lamel S, Qiao P, Sanborn JR, et al. Dermal
63. Burgess JL, Kovalchick DF, Siegel EM, Hysong TA, McCurdy SA.
exposure to methamphetamine hydrochloride contaminated
Medical surveil ance of clandestine drug laboratory investiga-
residential surfaces: surface pH values, volatility, and in vitro
tors. J Occup Environ Med 2002;44(2):184–9.
human skin. Food Chem Toxicol 2012;50(12):4436–40.
64. Czarnecki F. Chemical hazards in law enforcement. Clin Occup
47. Martyny JW, Arbuckle SL, McCammon CS, Esswein EJ, Erb N,
Environ Med 2003;3:443–56.
et al. Chemical concentrations and contamination associated
65. Witter RZ, Martyny JW, Mueller K, Gottschal B, Newman LS.
with clandestine methamphetamine laboratories. J Chem Health
Symptoms experienced by law enforcement personnel during
Safety 2007;14(4):40–52.
methamphetamine lab investigations. J Occup Environ Hyg
48. Martyny JW, Arbuckle SL, McCammon CS, Erb N. Methampheta-
2007;4(12):895–902.
mine contamination on environmental surfaces caused by
66. McCampbell MS. Meth and meth labs: the impact on sheriffs.
simulated smoking of methamphetamine. Denver, CO: National
Sheriff 2006;58(1):16–20.
Jewi sh Medical and Research Center, 2004.
67. Vanek M. Ten steps for EMS survival at clandestine metham-
49. Salocks CB. Assessment of children’s exposure to surface meth-
phetamine labs. Emerg Med Serv 2002;31(4):92, 6.
amphetamine residues in former clandestine methamphetamine 68. Burgess JL, Barnhart S, Checkoway H. Investigating clandestine
labs, and identification of a risk-based cleanup standard for sur-
drug laboratories: adverse medical effects in law enforcement
face methamphetamine contamination. Office of Environmental
personnel. Am J Ind Med 1996;30(4):488–94.
Health Hazard Assessment, Integrated Risk Assessment Branch, 69. Swetlow K. Children at clandestine methamphetamine labs:
2009.
helping meth’s youngest victims. US Department of Justice,
50. Martyny JW, Erb N, Arbuckle AL, VanDyke MV. A 24-hour study
Office of Justice Programs, 2003.
to investigate chemical exposures associated with clandestine
70. Mecham N, Melini J. Unintentional victims: development of a pro-
methamphetamine laboratories. Division of Environmental and
tocol for the care of children exposed to chemicals at metham-
Occupational Health Sciences, 2005.
phetamine laboratories. Pediatr Emerg Care 2002;18(4):327–32.
51. Gaynor K, Bevan M, Lee S, Swedenborg P. Clandestine meth-
71. Messina N, Marinelli-Casey P, West K, Rawson R. Children
amphetamine labs and wastes in Minnesota, Wipe Sampling,
exposed to methamphetamine use and manufacture. Child
Results, and Cleaning Former Meth Labs: Minnesota Studies’
Abuse Negl 2014;38(11):1872–83.
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
349
72. Land Levine S. Note: poison in our own backyards: what min-
97. O’Neil TB, Rawlins JM, Rea S, Wood FM. Methamphetamine
nesota legislators are doing to warn property purchasers of
laboratory-related burns in Western Australia–Why the explo-
the dangers of former clandestine methamphetamine labs.
sion? Burns 2011;37(6):1044–8.
Wm Mitchell L Rev 2005;31(4):1601–47.
98. Farst K, Duncan JM, Moss M, Ray RM, Kokoska E, et al. Meth-
73. Flannery MT, Jones J, Farst K, Worley KB, Worthington T, et al.
amphetamine exposure presenting as caustic ingestions in
The use of hair analysis to test children for exposure to metham-
children. Ann Emerg Med 2007;49(3):341–3.
phetamine. MSU J Med Law 2006;143:143–254.
99. Matteucci MJ, Auten JD, Crowley B, Combs D, Clark RF. Metham-
74. Manning T. Drug labs and endangered children. FBI Law Enforce-
phetamine exposures in young children. Pediatr Emerg Care
ment Bulletin 1999;68(7):10–14.
2007;23(9):638–40.
75. Denehy J. The meth epidemic: its effect on children and com-
100. Cline JS. Illegal methamphetamine laboratories as a public
munities. J Sch Nurs 2006;22(2):63–5.
health hazard. Pop Gov 2005:24–36.
76. Bratcher L, Wright Clayton E, Greeley C. Children in metham-
101. Rawson RA, Anglin MD, Ling W. Wil the methamphetamine
phetamine homes, a survey of physicians practicing in south-
problem go away? J Addict Dis 2002;21(1):5–19.
east Tennessee. Pediatr Emerg Care 2007;23(10):696–702.
102. Rothenbaum DK. Exposed: an officer’s story. CS Alert
77. Elmore L. Protection of children exposed to methamphetamine
2010;7(2):3–4.
production. Pop Gov 2005:28–30.
103. Hickey P. Police overcome by West Perth drug lab fumes Perth-
78. Department of Justice. Information bulletin, children at risk. U.S
Now. 13 April 2011.
Department of Justice, 2002.
104. Hickey P. Young cop struck down by clan lab fumes PerthNow.
79. Styles A. Chemical poisoning fears these school holidays WA
26 June 2011.
today. 8 July 2011.
105. Symonds K. Police officer hospitalised after drug lab bust
80. Jones L. Police concerns over amount of WA children forced to
PerthNow. 16 April 2011.
live in drug lab homes PerthNow. 9 July 2010.
106. Maxwell JC. Emerging research on methamphetamine. Curr
81. Melnikova N, Welles WL, Wilburn RE, Rice N, Wu J, et al. Hazards
Opin Psychiatry 2005;18(3):235–42.
of illicit methamphetamine production and efforts at reduction:
107. McKetin R, Hickey K, Devlin K, Lawrence K. The risk of psychotic
data from the hazardous substances emergency events surveil-
symptoms associated with recreational methamphetamine
lance system. Public Health Rep 2011;126(Suppl 1):116–23.
use. Drug Alcohol Rev 2010;29(4):358–63.
82. Jones L. Top cop tells of son’s drug lab trauma Sydney Morning
108. McKetin R, McLaren J, Lubman DI, Hides L. The prevalence of
Herald. 28 June 2011.
psychotic symptoms among methamphetamine users. Addic-
83. Cooper D. Acute public health consequences of methampheta-
tion 2006;101(10):1473–8.
mine laboratories–16 states, January 2000–June 2004. MMWR
109. McKetin R, Lubman DI, Baker AL, Dawe S, Ali RL. Dose-related
Morb Mortal Wkly Rep 2005;54(14):356–9.
psychotic symptoms in chronic methamphetamine users: evi-
84. Stewart F. ACT meth lab alert. The Canberra Times. 17 July 2011.
dence from a prospective longitudinal study. JAMA Psychiatry
85. Rose D. Meth labs pose toxic risk to community Sydney Morning
2013;70(3):319–24.
Herald. 26 August 2010.
110. Perez AY, Kirkpatrick MG, Gunderson EW, Marrone G, Silver
86. Hickey P. Police overcome by fumes from ‘Homeswest drug lab’
R, et al. Residual effects of intranasal methamphetamine
PerthNow. 21 June 2011.
on sleep, mood, and performance. Drug Alcohol Depend
87. O’Connell R, Knowles G. Neighbours report drug lab. The West
2008;94(1–3):258–62.
Australian. 2011 21 June 2011.
111. Ross GH, Sternquist MC. Methamphetamine exposure and
88. Oregon Department of Human Services. Children in Methamphet-
chronic illness in police officers: significant improvement
amine “Labs” In: Oregon. CD Summary, An Epdemiology Publica-
with sauna-based detoxification therapy. Toxicol Ind Health
tion of the Oregon Department of Human Services 2003;16(52):2.
2012;28(8):758–68.
89. Blostein P, Plaisier B, Maltz S, Davidson S, Wideman E, et al.
112. Haight W, Marshal J, Hans S, Black J, Sheridan K.
Methamphetamine production is hazardous to your health. J
“They mess with me, I mess with them”: understand-
Trauma 2009;66(6):1712–7.
ing physical aggression in rural girls and boys from
90. Horton DK, Berkowitz Z, Kaye WE. The acute health conse-
methamphetamine-involved families. Child Youth Serv Rev
quences to children exposed to hazardous substances used in
2010;32(10):1223–34.
illicit methamphetamine production, 1996 to 2001. J Childrens
113. Haight W, Jacobsen T, Black J, Kingery L, Sheridan K, et al.
Health 2003;1(1):99–108.
“In these bleak days”: parent methamphetamine abuse
91. Santos AP, Wilson AK, Hornung CA, Polk HC, Jr., Rodriguez JL,
and child welfare in the rural Midwest. Child Youth Serv Rev
et al. Methamphetamine laboratory explosions: a new and
2005;27(8):949–71.
emerging burn injury. J Burn Care Rehabil 2005;26(3):228–32.
114. Haight W, Black J, Sheridan K. A mental health intervention for
92. Symonds K. Man, 48, in intensive care after Millendon drug lab
rural, Foster children from methamphetamine-involved fami-
fire PerthNow. 12 August 2011.
lies: experimental assessment with qualitative elaboration.
93. Hickey P. Commissioner Karl O’Cal aghan’s son hurt in ‘drug lab’
Child Youth Serv Rev 2010;32(10):1146–457.
blast. PerthNow. 21 March 2011.
115. Ostler T, Haight W, Black J, Choi GY, Kingery L, et al. Case
94. Hickey P. Armadale blast leads to 100th clandestine drug lab
series: mental health needs and perspectives of rural children
PerthNow. 6 July 2011.
reared by parents who abuse methamphetamine. J Am Acad
95. Robinson G. Sydney ‘lab’ blast: burnt man arrested Sydney
Child Adolesc Psychiatry 2007;46(4):500–7.
Morning Herald. 15 March 2010.
116. Asanbe C, Hal C, Bolden C. The methamphetamine home: psy-
96. Robinson C, Hickey P. ’Drug lab’ explosion in Gosnells blows
chological impact on preschoolers in rural Tennessee. J Rural
roof off house PerthNow. 3 June 2011.
Health 2008;24(3):229–34.
Unauthenticated
Download Date | 10/11/16 7:20 AM
350 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
117. Hohman M, Oliver R, Wright W. Methamphetamine abuse
methamphetamine-involved families: implications for rural
and manufacture: the child welfare response. Soc Work
child welfare practice. J Public Child Welf 2014;8(5):514–38.
2004;49(3):373–81.
136. Messina N, Jeter K, Marinelli-Casey P, West K, Rawson R.
118. Zernike K. A drug scourge creates its own form of orphan. The
Children exposed to methamphetamine use and manufacture.
New York Times. 11 July 2005.
Child Abuse Negl 2014;38(11):1872–83.
119. Walsh C, MacMil an HL, Jamieson E. The relationship between
137. Oral R, Bayman L, Assad A, Wibbenmeyer L, Buhrow J, et al.
parental substance abuse and child maltreatment: find-
Illicit drug exposure in patients evaluated for alleged child
ings from the Ontario health supplement. Child Abuse Negl
abuse and neglect. Pediatr Emerg Care 2011;27(6):490–5.
2003;27(12):1409–25.
138. enHealth. Environmental health risk assessment, guidelines
120. Osborne C, Berger LM. Parental substance abuse and child
for assessing human health risks from environmental hazards.
well-being, a consideration of parents’ gender and coresi-
Canberra: Commonwealth of Australia, 2012 ISBN: 978-1-
dence. J Family Issues 2009;30(3):341–70.
74241-766-0.
121. LaGasse LL, Derauf C, Smith LM, Newman E, Shah R,
139. Ministry of Health. Guidelines for the remediation of clan-
et al. Prenatal methamphetamine exposure and child-
destine methamphetamine laboratory sites. Wellington: New
hood behavior problems at 3 and 5 years of age. Pediatrics
Zealand Ministry of Health, 2010.
2012;129(4):681–8.
140. USEPA. Voluntary guidelines for methamphetamine laboratory
122. Billing L, Eriksson M, Larsson G, Zetterstrom R. Amphetamine
cleanup. U.S. Environmental Protection Agency, 2009.
addiction and pregnancy. III. One year follow-up of the chil-
141. Rusnal SM, Ginsberg G, Toal B. Guidelines for the cleanup of
dren. Psychosocial and pediatric aspects. Acta Paediatr Scand
Connecticut methamphetamine labs. Connecticut: Depart-
1980;69(5):675–80.
ment of Public Health, Environmental and Occupational Health
123. Billing L, Eriksson M, Steneroth G, Zetterstrom R. Pre-school
Assessment Program, 2006.
children of amphetamine-addicted mothers. I. Somatic
142. Alaska Department of Environmental Conservation. Guid-
and psychomotor development. Acta Paediatr Scand
ance and standards for cleanup of illegal drug-manufacturing
1985;74(2):179–84.
sites. Alaska Department of Environmental Conservation, Spil
124. Eriksson M, Billing L, Steneroth G, Zetterstrom R. Health
Prevention and Response Division, Prevention and Emergency
and development of 8-year-old children whose mothers
Response Program, 2007.
abused amphetamine during pregnancy. Acta Paediatr Scand
143. Colorado Department of Public Health and Environment.
1989;78(6):944–9.
Cleanup of clandestine methamphetamine labs guidance docu-
125. Billing L, Eriksson M, Jonsson B, Steneroth G, Zetterstrom R.
ment. Hazardous Materials and Waste Management Division,
The influence of environmental factors on behavioural prob-
State of Colorado, 2007.
lems in 8-year-old children exposed to amphetamine during
144. Colorado Department of Public Health and Environment. Sup-
fetal life. Child Abuse Negl 1994;18(1):3–9.
port for selection of a cleanup level for methamphetamine at
126. Cernerud L, Eriksson M, Jonsson B, Steneroth G, Zetter-
clandestine drug laboratories. State of Colorado, 2005.
strom R. Amphetamine addiction during pregnancy: 14-year
145. Kentucky Department for Environment Protection. Kentucky
follow-up of growth and school performance. Acta Paediatr
cleanup guidance for methamphetamine contaminated proper-
1996;85(2):204–8.
ties. Energy and Environment, Department for Environmental
127. Smith LM, LaGasse LL, Derauf C, Newman E, Shah R, et al.
Protection, Division of Waste Management, 2009.
Motor and cognitive outcomes through three years of age in
146. Michigan Department of Community Health. Cleanup of clan-
children exposed to prenatal methamphetamine. Neurotoxicol
destine drug laboratory guidance. Michigan: Department of
Teratol 2011;33(1):176–84.
Community Health, 2007.
128. Smith LM, Chang L, Yonekura ML, Grob C, Osborn D, et al. Brain 147. Minnesota Department of Health. Clandestine drug lab general
proton magnetic resonance spectroscopy in children exposed
cleanup guidance. Minnesota: Department of Health Division of
to methamphetamine in utero. Neurology 2001;57(2):255–60.
Environmental Health and Minnesota Pollution Control Agency,
129. Siegel JA, Park BS, Raber J. Methamphetamine exposure during
2010.
brain development alters the brain acetylcholine system in
148. North Carolina Department of Health and Human Services.
adolescent mice. J Neurochem 2011;119(1):89–99.
Illegal methamphetamine laboratory decontamination and
130. Siegel JA, Park BS, Raber J. Long-term effects of neonatal meth-
re-occupancy guidelines. State of Northern Carolina: Depart-
amphetamine exposure on cognitive function in adolescent
ment of Health and Human Services, Division of Public Health,
mice. Behav Brain Res 2011;219(1):159–64.
Occupational and Environmental Epidemiology Branch, 2005.
131. North A, Swant J, Salvatore MF, Gamble-George J, Prins P, et al.
149. Washington State Department of Health. Guidelines for envi-
Chronic methamphetamine exposure produces a delayed,
ronmental sampling at illegal drug manufacturing sites. Wash-
long-lasting memory deficit. Synapse 2013;67(5):245–57.
ington State Department of Health, Division of Environmental
132. Wells K. Substance abuse and child maltreatment. Pediatr Clin
Health, 2005.
North Am 2009;56(2):345–62.
150. County of Stanislaus. Criteria for the assessment and remedia-
133. Keltner L, Chervenak C, Tsongas T. Clandestine methampheta-
tion of methamphetamine laboratories. Department of Environ-
mine labs: risks to children. Epidemiology 2004;15(4):S88.
mental Resources, 2007.
134. Messina N, Jeter K. Parental methamphetamine use and
151. Hammon TL, Griffin S. Support for selection of a methampheta-
manufacture: child and familial outcomes. J Public Child Welf
mine cleanup standard in Colorado. Regul Toxicol Pharmacol
2012;6(3):296–312.
2007;48(1):102–14.
135. Sheridan K. A systematic review of the literature regard-
152. Salocks C, Golub MS, Kaufman FL. Development of a reference
ing family context and mental health of children from rural
dose (RfD) for methamphetamine. Office of Environmental
Unauthenticated
Download Date | 10/11/16 7:20 AM
Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
351
Health Hazard Assessment, Integrated Risk Assessment Branch, 169. Cook CE, Jeffcoat AR, Hil JM, Pugh DE, Patetta PK, et al.
2009.
Pharmacokinetics of methamphetamine self-administered to
153. Cohen Hubal EA, Sheldon LS, Burke JM, McCurdy TR, Berry
human subjects by smoking S-(+)-methamphetamine hydro-
MR, et al. Children’s exposure assessment: a review of factors
chloride. Drug Metab Dispos 1993;21(4):717–23.
influencing Children’s exposure, and the data available to
170. Golub M, Costa L, Crofton K, Frank D, Fried P, et al. NTP-CERHR
characterize and assess that exposure. Environ Health Perspect
expert panel report on the reproductive and developmental
2000;108(6):475–86.
toxicity of amphetamine and methamphetamine. Birth Defects
154. Salocks CB, Hui X, Lamel S, Hafeez F, Qiao P, et al. Dermal
Res B Dev Reprod Toxicol 2005;74(6):471–584.
exposure to methamphetamine hydrochloride contaminated
171. Cook CE, Jeffcoat AR, Sadler BM, Hil JM, Voyksner RD, et al.
residential surfaces II. Skin surface contact and dermal transfer
Pharmacokinetics of oral methamphetamine and effects
relationship. Food Chem Toxicol 2014;66:1–6.
of repeated daily dosing in humans. Drug Metab Dispos
155. Parker K, Morrison G. Methamphetamine absorption by skin
1992;20(6):856–62.
lipids: accumulated mass, partition coefficients, and the influ-
172. Li L, Lopez JC, Galloway GP, Baggott MJ, Everhart T, et al.
ence of fatty acids. Indoor Air 2015.
Estimating the intake of abused methamphetamines using
156. Morrison G, Shakila NV, Parker K. Accumulation of gas-phase
experimenter-administered deuterium labeled R-methamphet-
methamphetamine on clothing, toy fabrics, and skin oil. Indoor
amine: selection of the R-methamphetamine dose. Ther Drug
Air 2015;25(4):405–14.
Monit 2010;32(4):504–7.
157. Patrick G, Daniell W, Treser C. Residual methamphetamine in
173. Franksson G, Anggard E. The plasma protein binding of
decontaminated clandestine drug laboratories. J Occup Environ
amphetamine, catecholamines and related compounds. Acta
Hyg 2009;6(3):151–6.
Pharmacol Toxicol (Copenh) 1970;28(3):209–14.
158. Burton BT. Heavy metal and organic contaminants associated
174. de la Torre R, Farre M, Navarro M, Pacifici R, Zuccaro P, et al.
with illicit methamphetamine production. NIDA Res Monogr
Clinical pharmacokinetics of amfetamine and related sub-
1991;115:47–59.
stances: monitoring in conventional and non-conventional
159. Man G, Stoeber B, Walus K. An assessment of sensing tech-
matrices. Clin Pharmacokinet 2004;43(3):157–85.
nologies for the detection of clandestine methamphetamine
175. Steiner E, Villen T, Hal berg M, Rane A. Amphetamine secretion
drug laboratories. Forensic Sci Int 2009;189(1–3):1–13.
in breast milk. Eur J Clin Pharmacol 1984;27(1):123–4.
160. Abdul ah AF, Miskelly GM. Recoveries of trace pseudoephed-
176. Kraemer T, Maurer HH. Toxicokinetics of amphetamines: metab-
rine and methamphetamine residues from impermeable house-
olism and toxicokinetic data of designer drugs, amphetamine,
hold surfaces: implications for sampling methods used during
methamphetamine, and their N-alkyl derivatives. Ther Drug
remediation of clandestine methamphetamine laboratories.
Monit 2002;24(2):277–89.
Talanta 2010;81(1–2):455–61.
177. Cruickshank CC, Dyer KR. A review of the clinical pharmacology
161. NIOSH. 9106 Methamphetamine and illicit drugs, precursors,
of methamphetamine. Addiction 2009;104(7):1085–99.
and adulterants on wipes by liquid-liquid extraction. NIOSH
178. Jones AW, Holmgren A. Concentration ratios of methampheta-
manual of analytical methods (NMAM), 5th ed. CDC, The
mine to amphetamine in blood can help to distinguish use of
National Institute for Occupational Safety and Health, 2011.
methamphetamine from various mixtures of the two stimu-
162. NIOSH. Method 9109, methamphetamine and illicit drugs, pre-
lants. J Anal Toxicol 2012;36(9):634–7.
cursors, and adulterants on wipes by solid phase extraction.
179. Cone EJ. Saliva testing for drugs of abuse. Ann N Y Acad Sci
NIOSH manual of analytical methods (NMAM), 5th ed. CDC, The
1993;694:91–127.
National Institute for Occupational Safety and Health, 2011.
180. Barnes AJ, Smith ML, Kacinko SL, Schwilke EW, Cone EJ, et al.
163. NIOSH. Method 9111 methamphetamine on wipes by liquid
Excretion of methamphetamine and amphetamine in human
chromatography-mass spectrometry-SIM NIOSH manual of
sweat following controlled oral methamphetamine administra-
analytical methods (NMAM), 5th ed. CDC, The National Institute
tion. Clin Chem 2008;54(1):172–80.
for Occupational Safety and Health, 2011.
181. Schepers RJ, Oyler JM, Joseph RE Jr, Cone EJ, Moolchan ET,
164. NIOSH. NIOSH manual of analytical methods (NMAM), 5th
et al. Methamphetamine and amphetamine pharmacokinet-
ed. Washington: CDC, The National Institute for Occupational
ics in oral fluid and plasma after controlled oral metham-
Safety and Health, 2016.
phetamine administration to human volunteers. Clin Chem
165. SKC. MethChek immunoassay wipe kit for methamphetamine
2003;49(1):121–32.
residue on surfaces and performance of methchek immunoas-
182. Huang MC, Chang BL, Liao CH, Liu RH. Drugs of abuse, urine.
say wipe kits. 2009.
Academic Press, 2000:651–62.
166. Grange AH, Sovocool GW. Detection of illicit drugs on
183. Oyler JM, Cone EJ, Joseph RE, Jr., Moolchan ET, Huestis MA.
surfaces using direct analysis in real time (DART) time-of-
Duration of detectable methamphetamine and amphetamine
flight mass spectrometry. Rapid Commun Mass Spectrom
excretion in urine after controlled oral administration of meth-
2011;25(9):1271–81.
amphetamine to humans. Clinical Chem 2002;48(10):1703–14.
167. Van Dyke MV, Serrano KA, Kofford S, Contreras J, Martyny
184. Musshoff F. Illegal or legitimate use? Precursor compounds
JW. Variability and specificity associated with environmental
to amphetamine and methamphetamine. Drug Metab Rev
methamphetamine sampling and analysis. J Occup Environ Hyg
2000;32(1):15–44.
2011;8(11):636–41.
185. Jusko WJ, Milsap RL. Pharmacokinetic principles of drug distri-
168. Harris DS, Boxenbaum H, Everhart ET, Sequeira G, Mendelson
bution in saliva. Ann N Y Acad Sci 1993;694:36–47.
JE, et al. The bioavailability of intranasal and smoked metham-
186. Barnes AJ, De Martinis BS, Gorelick DA, Goodwin RS,
phetamine. Clin Pharmacol Ther 2003;74(5):475–86.
Kolbrich EA, et al. Disposition of MDMA and metabolites in
Unauthenticated
Download Date | 10/11/16 7:20 AM
352 Wright et al.: Exposures associated with clandestine methamphetamine drug laboratories in Australia
human sweat following controlled MDMA administration. Clini-
203. Han E, Park Y, Kim E, Lee S, Choi H, et al. The dependence of
cal Chem 2009;55(3):454–62.
the incorporation of methamphetamine into rat hair on dose,
187. Kidwell DA, Smith FP. Susceptibility of PharmChek drugs of
frequency of administration and hair pigmentation. J Chroma-
abuse patch to environmental contamination. Forensic Sci Int
togr B Analyt Technol Biomed Life Sci 2010;878(28):2845–51.
2001;116(2–3):89–106.
204. Han E, Paulus MP, Wittmann M, Chung H, Song JM. Hair analy-
188. Lin DL, Yin RM, Liu HC, Wang CY, Liu RH. Deposition charac-
sis and self-report of methamphetamine use by methampheta-
teristics of methamphetamine and amphetamine in fingernail
mine dependent individuals. J Chromatogr B Analyt Technol
clippings and hair sections. J Anal Toxicol 2004;28(6):411–7.
Biomed Life Sci 2011;879(7–8):541–7.
189. Suzuki O, Hattori H, Asano M. Nails as useful materials for
205. Boroda A, Gray W. Hair analysis for drugs in child abuse. J R Soc
detection of methamphetamine or amphetamine abuse. Foren-
Med 2005;98(7):318–9.
sic Sci Int 1984;24(1):9–16.
206. Lewis D, Moore C, Morrissey P, Leikin J. Determination of drug
190. Cooper GAA. Chapter 1 – Anatomy and Physiology of Hair, and
exposure using hair: application to child protective cases.
Principles for its Collection. In: Vincenti PKS, editor. Hair analy-
Forensic Sci Int 1997;84(1–3):123–8.
sis in clinical and forensic toxicology. Boston: Academic Press,
207. Bassindale T. Quantitative analysis of methamphetamine in hair
2015:1–22.
of children removed from clandestine laboratories–evidence of
191. Mieczkowski T. Hair analysis. Substance Misuse: Elsevier Ltd,
passive exposure? Forensic Sci Int 2012;219(1–3):179–82.
2005:183–92.
208. Moller M, Koren G, Karaskov T, Garcia-Bournissen F. Examining
192. Nakahara Y. Detection and diagnostic interpretation of
the health and drug exposures among Canadian children resid-
amphetamines in hair. Forensic Sci Int 1995;70(1–3):135–53.
ing in drug-producing homes. J Pediatr 2011;159(5):766–70.
193. Klein J, Karaskov T, Koren G. Clinical applications of hair testing 209. Nakahara Y, Takahashi K, Kikura R. Hair analysis for drugs of
for drugs of abuse–the Canadian experience. Forensic Sci Int
abuse. X. Effect of physicochemical properties of drugs on the
2000;107(1–3):281–8.
incorporation rates into hair. Biol Pharm Bul 1995;18(9):1223–7.
194. Stout PR, Ropero-Miller JD, Baylor MR, Mitchell JM. Morpho-
210. Kintz P, Cirimele V, Tracqui A, Mangin P. Simultaneous determi-
logical changes in human head hair subjected to various
nation of amphetamine, methamphetamine, 3,4-methylenedi-
drug testing decontamination strategies. Forensic Sci Int
oxyamphetamine and 3,4-methylenedioxymethamphetamine
2007;172(2–3):164–70.
in human hair by gas chromatography-mass spectrometry. J
195. Boumba VA, Ziavrou KS, Vougiouklakis T. Hair as a biological
Chromatogr B Biomed Appl 1995;670(1):162–6.
indicator of drug use, drug abuse or chronic exposure to envi-
211. Rohrich J, Kauert G. Determination of amphetamine and
ronmental toxicants. Int J Toxicol 2006;25(3):143–63.
methylenedioxy-amphetamine-derivatives in hair. Forensic Sci
196. Farst K, Reading Meyer JA, Mac Bird T, James L, Robbins
Int 1997;84(1–3):179–88.
JM. Hair drug testing of children suspected of exposure to
212. Lin DL, Yin RM, Liu RH. Gas chromatography-mass spectrom-
the manufacture of methamphetamine. J Forensic Leg Med
etry (GC-MS) analysis of amphetamine, methamphetamine,
2011;18(3):110–4.
3,4-methylenedioxyamphetamine and 3,4-methylenedioxym-
197. Nakahara Y, Kikura R, Takahashi K. Hair analysis for
ethamphetamine in human hair and hair Sections. J Food Drug
drugs of abuse XX. Incorporation and behaviors of seven
Anal 2005;13(3):193–200.
methamphetamine homologs in the rat hair root. Life Sci
213. Miyaguchi H, Iwata YT, Kanamori T, Tsujikawa K, Kuwayama
1998;63(10):883–93.
K, et al. Rapid identification and quantification of metham-
198. Nakahara Y, Kikura R. Hair analysis for drugs of abuse. XIII.
phetamine and amphetamine in hair by gas chromatography/
Effect of structural factors on incorporation of drugs into hair:
mass spectrometry coupled with micropulverized extraction,
the incorporation rates of amphetamine analogs. Arch Toxicol
aqueous acetylation and microextraction by packed sorbent. J
1996;70(12):841–9.
Chromatogr A 2009;1216(18):4063–70.
199. Polettini A, Cone EJ, Gorelick DA, Huestis MA. Incorporation of
214. Miyaguchi H, Takahashi H, Ohashi T, Mawatari K, Iwata YT,
methamphetamine and amphetamine in human hair following
et al. Rapid analysis of methamphetamine in hair by microp-
controlled oral methamphetamine administration. Anal Chim
ulverized extraction and microchip-based competitive ELISA.
Acta 2012;726:35–43.
Forensic Sci Int 2009;184(1–3):1–5.
200. Baumgartner W, Hil V. Hair analysis for organic analytes: meth-
215. Meng P, Fang N, Wang M, Liu H, Chen DD. Analysis of
odology, reliability issues, and field studies. In: Kintz P, editor.
amphetamine, methamphetamine and methylenedioxy-
CRC Press, 1996.
methamphetamine by micellar capil ary electrophoresis
201. Williams J, Patsalos PN, Wilson JF. Hair analysis as a potential
using cation-selective exhaustive injection. Electrophoresis
index of therapeutic compliance in the treatment of epilepsy.
2006;27(16):3210–7.
Forensic Sci Int 1997;84(1–3):113–22.
216. Kelly RC, Mieczkowski T, Sweeney SA, Bourland JA. Hair
202. Williams J. The assessment of therapeutic compliance based on
analysis for drugs of abuse. Hair color and race differentials
the analysis of drug concentrations in hair. In: Mieczkowski T,
or systematic differences in drug preferences? Forensic Sci Int
editor. CRC Press, 1999.
2000;107(1–3):63–86.
Unauthenticated
Download Date | 10/11/16 7:20 AM




